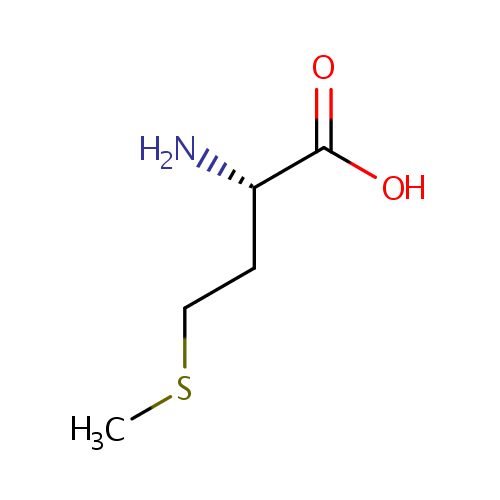
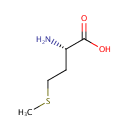L-Methionine (PAMDB000175)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000175 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | L-Methionine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | L-Methionine, in addition to being a substrate for protein synthesis, is an intermediate in transmethylation reactions, serving as the major methyl group donor, including the methyl groups for DNA and RNA intermediates. Methionine is a methyl acceptor for 5-methyltetrahydrofolate-homocysteine methyl transferase (methionine synthase), the only reaction that allows for the recycling of this form of folate, and is also a methyl acceptor for the catabolism of betaine. Methionine is also required for synthesis of cysteine. Methionine is accepted as the metabolic precursor for cysteine. Only the sulfur atom from methionine is transferred to cysteine; the carbon skeleton of cysteine is donated by serine. (PMID 16702340) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C5H11NO2S | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 149.211 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 149.051049291 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | FFEARJCKVFRZRR-BYPYZUCNSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C5H11NO2S/c1-9-3-2-4(6)5(7)8/h4H,2-3,6H2,1H3,(H,7,8)/t4-/m0/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 63-68-3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | (2S)-2-amino-4-(methylsulfanyl)butanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | L-methionine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | CSCC[C@H](N)C(O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as d-alpha-amino acids. These are alpha amino acids which have the D-configuration of the alpha-carbon atom. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carboxylic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Amino acids, peptides, and analogues | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | D-alpha-amino acids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | 284 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | Methionine sulfoxide + Reduced Thioredoxin > Water + L-Methionine + Oxidized Thioredoxin Adenosine triphosphate + Water + L-Methionine > ADP + Hydrogen ion + L-Methionine + Phosphate Adenosine triphosphate + Water + L-Methionine > ADP + Hydrogen ion + L-Methionine + Phosphate 5-Methyltetrahydrofolic acid + L-Homocysteine <> Hydrogen ion + L-Methionine + Tetrahydrofolic acid L-Homocysteine + S-Methylmethionine > Hydrogen ion +2 L-Methionine S-Adenosylmethionine + L-Homocysteine + S-Methylmethionine <> S-Adenosylhomocysteine + Hydrogen ion + L-Methionine [4Fe-4S] iron-sulfur cluster + 2 S-Adenosylmethionine + Hydrogen ion + NAD + octanoate (protein bound) > [2Fe-2S] iron-sulfur cluster +2 5'-Deoxyadenosine +2 Iron + lipoate (protein bound) +2 L-Methionine + NADH [2Fe-2S] iron-sulfur cluster + S-Adenosylmethionine + Dethiobiotin > [2Fe-1S] desulfurated iron-sulfur cluster + Biotin + 5'-Deoxyadenosine + Hydrogen ion + L-Methionine Adenosine triphosphate + L-Methionine + tRNA(Met) > Adenosine monophosphate + L-Methionyl-tRNA (Met) + Pyrophosphate Adenosine triphosphate + Water + L-Methionine <> S-Adenosylmethionine + Phosphate + Pyrophosphate 2 S-Adenosylmethionine + Coproporphyrin III <>2 Carbon dioxide +2 5'-Deoxyadenosine +2 L-Methionine + Protoporphyrinogen IX S-Adenosylmethionine + NADPH + L-Tyrosine > p-Cresol + 5'-Deoxyadenosine + Dehydroglycine + Hydrogen ion + L-Methionine + NADP Hydrogen peroxide + L-Methionine > Water + Methionine sulfoxide S-Adenosylmethionine + L-Homocysteine <> S-Adenosylhomocysteine + L-Methionine 5-Methyltetrahydrofolic acid + L-Homocysteine <> Tetrahydrofolic acid + L-Methionine Dethiobiotin + Sulfur donor + 2 S-Adenosylmethionine + 2 e- + 2 Hydrogen ion <> Biotin +2 L-Methionine +2 5'-Deoxyadenosine 4-Amino-5-hydroxymethyl-2-methylpyrimidine + S-Adenosylmethionine <> 5-Aminoimidazole ribonucleotide + 4-Amino-2-methyl-5-phosphomethylpyrimidine + 5'-Deoxyadenosine + L-Methionine + Formic acid + CO Adenosine triphosphate + L-Methionine + tRNA(Met) + tRNA(Met) <> Adenosine monophosphate + Pyrophosphate + L-Methionyl-tRNA + L-Methionyl-tRNA 5-Methyltetrahydropteroyltri-L-glutamic acid + L-Homocysteine <> Tetrahydropteroyltri-L-glutamic acid + L-Methionine 2-Oxo-4-methylthiobutanoic acid + L-Glutamate <> L-Methionine + alpha-Ketoglutarate Protein N6-(octanoyl)lysine + 2 Sulfur donor + 2 S-Adenosylmethionine + Protein N6-(octanoyl)lysine <> Protein N6-(lipoyl)lysine +2 L-Methionine +2 5'-Deoxyadenosine + Protein N6-(lipoyl)lysine Octanoyl-[acp] + 2 Sulfur donor + 2 S-Adenosylmethionine <> Lipoyl-[acp] +2 L-Methionine +2 5'-Deoxyadenosine More...L-Methionine + Hydrogen peroxide > L-methionine <i>S</i>-oxide + Water Hydrogen ion + α-D-ribose-1-methylphosphonate-5-phosphate + S-Adenosylmethionine > α-D-ribose-1,2-cyclic-phosphate-5-phosphate + methane + 5'-Deoxyadenosine + L-Methionine L-Methionine + Acetyl-CoA N-α-acetyl-L-methionine + Coenzyme A <i>S</i>-sulfanyl-[acceptor] + Dethiobiotin + S-Adenosylmethionine > an unsulfurated sulfur acceptor + Biotin + 5'-Deoxyadenosine + L-Methionine + Hydrogen ion a protein with N-terminal methionine + Water > L-Methionine + Peptides Coproporphyrinogen III + S-Adenosylmethionine > Protoporphyrinogen IX + Carbon dioxide + L-Methionine + 5'-Deoxyadenosine L-Homocysteine + 5-Methyltetrahydropteroyltri-L-glutamic acid > L-Methionine + tetrahydropteroyl tri-L-glutamate L-Homocysteine + S-Adenosylmethionine Hydrogen ion + L-Methionine + S-Adenosylhomocysteine S-methyl-L-methionine + L-Homocysteine Hydrogen ion + L-Methionine 5-Aminoimidazole ribonucleotide + S-Adenosylmethionine 4-Amino-2-methyl-5-phosphomethylpyrimidine + 5'-Deoxyadenosine + L-Methionine + Formic acid + carbon monoxide + Hydrogen ion L-Methionine + a 2-oxo carboxylate 2-Oxo-4-methylthiobutanoic acid + a standard α amino acid S-Adenosylmethionine + Ribonuc-tri-P-reductases-inactive <> 5'-Deoxyadenosine + L-Methionine + Ribonuc-tri-P-reductases-active L-Tyrosine + S-Adenosylmethionine + a reduced electron acceptor > Dehydroglycine + p-Cresol + 5'-Deoxyadenosine + L-Methionine + an oxidized electron acceptor + Hydrogen ion -->-->N-6-isopentyl adenosine-37 tRNA + S-Adenosylmethionine + <i>S</i>-sulfanyl-[acceptor] 2-methylthio-N-6-isopentyl adenosine-37 tRNA + S-Adenosylhomocysteine + L-Methionine + 5'-Deoxyadenosine + an unsulfurated sulfur acceptor + Hydrogen ion 6-Carboxy-5,6,7,8-tetrahydropterin + S-Adenosylmethionine + Hydrogen ion > 7-carboxy-7-deazaguanine + 5'-Deoxyadenosine + L-Methionine + Ammonia gly-met + Water > Glycine + L-Methionine methionine-alanine dipeptide + Water > L-Methionine + L-Alanine 5-methyltetrahydropteroyltri-L-glutamate + L-Homocysteine > tetrahydropteroyltri-L-glutamate + L-Methionine L-Methionine + thioredoxin disulfide + Water > L-methionine (S)-S-oxide + thioredoxin L-Methionine + thioredoxin disulfide + Water > L-Methionine (R)-S-oxide + thioredoxin Adenosine triphosphate + L-Methionine + tRNA(Met) > Adenosine monophosphate + Pyrophosphate + L-methionyl-tRNA(Met) L-Methionine + a 2-oxo acid > 2-Oxo-4-methylthiobutanoic acid + an L-amino acid 2 S-Adenosylmethionine <> S-Adenosylhomocysteine +2 L-Methionine + 5'-Deoxyadenosine 2 S-Adenosylmethionine + Reduced acceptor <> S-Adenosylhomocysteine +2 L-Methionine + 5'-Deoxyadenosine L-Tyrosine + S-Adenosylmethionine + NADPH <> 2-iminoacetate + p-Cresol + 5'-Deoxyadenosine + L-Methionine + NADP + Hydrogen ion Peptide-L-methionine + Thioredoxin disulfide + Water + L-Methionine <> Peptide-L-methionine (S)-S-oxide + Thioredoxin + L-methionine (S)-S-oxide Dethiobiotin + 2 S-adenosyl-L-methionine + 2 Hydrogen ion + a sulfurated [sulfur carrier] > Biotin +2 L-Methionine +2 5'-Deoxyadenosine Octanoyl-[acyl-carrier protein] + 2 a sulfur donor + 2 S-adenosyl-L-methionine > Lipoyl-ACP +2 L-Methionine + 5'-Deoxyadenosine Protein N6-(octanoyl)lysine + 2 a sulfur donor + 2 S-adenosyl-L-methionine > Protein N6-(lipoyl)lysine +2 L-Methionine +2 5'-Deoxyadenosine Protein N6-(octanoyl)lysine + 2 Reduced ferredoxin + 2 a sulfurated [sulfur carrier]? + 2 S-adenosyl-L-methionine >2 L-Methionine +2 5'-Deoxyadenosine + Oxidized ferredoxin + Protein N6-(lipoyl)lysine + an unsulfurated [sulfur carrier] 5-Methyltetrahydrofolic acid + Homocysteine + 5-Methyltetrahydrofolic acid + Homocysteine > Tetrahydrofolic acid + L-Methionine + Tetrahydrofolic acid Homocysteine + N5-methyl--tetrahydropteroyl tri-L-glutamate + Homocysteine > L-Methionine + tetrahydropteroyltri-L-glutamate L-Methionine + Adenosine triphosphate + Hydrogen ion + tRNA(Met) > Adenosine monophosphate + Pyrophosphate + L-methionyl-tRNA(Met) Homocysteine + N5-methyl--tetrahydropteroyl tri-L-glutamate + Homocysteine > tetrahydropteroyltri-L-glutamate + L-Methionine L-Methionine + Water + Adenosine triphosphate > Phosphate + Pyrophosphate L-Methionine + Water + Adenosine triphosphate > Phosphate + Pyrophosphate + S-adenosyl-L-methionine S-adenosyl-L-methionine + Coproporphyrinogen III > 5'-Deoxyadenosine + L-Methionine + Carbon dioxide + Protoporphyrinogen IX L-Tyrosine + NADPH + S-adenosyl-L-methionine + L-Tyrosine + NADPH > Hydrogen ion + NADP + L-Methionine + 5'-Deoxyadenosine + p-Cresol + 2-iminoacetate 7-aminomethyl-7-deazaguanosine34 in tRNA + S-adenosyl-L-methionine > Hydrogen ion + L-Methionine + Adenine + epoxyqueuosine L-Methionine + Adenosine triphosphate + Water > Adenosine diphosphate + Phosphate + Hydrogen ion + L-Methionine + ADP L-Methionine + Adenosine triphosphate + Water > Adenosine diphosphate + Phosphate + Hydrogen ion + L-Methionine + ADP L-Methionine + Adenosine triphosphate + Water > Adenosine diphosphate + Pyrophosphate + Hydrogen ion + L-Methionine + ADP L-Methionine + Adenosine triphosphate + Water > Adenosine diphosphate + Pyrophosphate + Hydrogen ion + L-Methionine + ADP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Boy, Matthias; Klein, Daniela; Schroeder, Hartwig. Method for the production and recovery of methionine. PCT Int. Appl. (2005), 34 pp. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in nucleotide binding
- Specific function:
- Is required not only for elongation of protein synthesis but also for the initiation of all mRNA translation through initiator tRNA(fMet) aminoacylation
- Gene Name:
- metG
- Locus Tag:
- PA3482
- Molecular weight:
- 74.9 kDa
Reactions
| ATP + L-methionine + tRNA(Met) = AMP + diphosphate + L-methionyl-tRNA(Met). |
- General function:
- Involved in transferase activity
- Specific function:
- An aromatic amino acid + 2-oxoglutarate = an aromatic oxo acid + L-glutamate
- Gene Name:
- tyrB
- Locus Tag:
- PA3139
- Molecular weight:
- 43.3 kDa
Reactions
| An aromatic amino acid + 2-oxoglutarate = an aromatic oxo acid + L-glutamate. |
- General function:
- Involved in oxidoreductase activity, acting on a sulfur group of donors, disulfide as acceptor
- Specific function:
- Could have an important function as a repair enzyme for proteins that have been inactivated by oxidation. Catalyzes the reversible oxidation-reduction of methionine sulfoxide in proteins to methionine
- Gene Name:
- msrA
- Locus Tag:
- PA5018
- Molecular weight:
- 23.5 kDa
Reactions
| Peptide-L-methionine + thioredoxin disulfide + H(2)O = peptide-L-methionine (S)-S-oxide + thioredoxin. |
| L-methionine + thioredoxin disulfide + H(2)O = L-methionine (S)-S-oxide + thioredoxin. |
- General function:
- Involved in peptide-methionine-(S)-S-oxide reductase activity
- Specific function:
- Peptide-L-methionine + thioredoxin disulfide + H(2)O = peptide-L-methionine (R)-S-oxide + thioredoxin
- Gene Name:
- msrB
- Locus Tag:
- PA2827
- Molecular weight:
- 14.8 kDa
Reactions
| Peptide-L-methionine + thioredoxin disulfide + H(2)O = peptide-L-methionine (R)-S-oxide + thioredoxin. |
- General function:
- Involved in methionine adenosyltransferase activity
- Specific function:
- Catalyzes the formation of S-adenosylmethionine from methionine and ATP. The overall synthetic reaction is composed of two sequential steps, AdoMet formation and the subsequent tripolyphosphate hydrolysis which occurs prior to release of AdoMet from the enzyme. Is essential for growth
- Gene Name:
- metK
- Locus Tag:
- PA0546
- Molecular weight:
- 42.7 kDa
Reactions
| ATP + L-methionine + H(2)O = phosphate + diphosphate + S-adenosyl-L-methionine. |
- General function:
- Involved in [formate-C-acetyltransferase]-activating enzyme activity
- Specific function:
- Activation of anaerobic ribonucleoside-triphosphate reductase under anaerobic conditions by generation of an organic free radical, using S-adenosylmethionine and reduced flavodoxin as cosubstrates to produce 5'-deoxy-adenosine
- Gene Name:
- nrdG
- Locus Tag:
- PA1919
- Molecular weight:
- 25.7 kDa
- General function:
- Involved in catalytic activity
- Specific function:
- Catalyzes the conversion of dethiobiotin (DTB) to biotin by the insertion of a sulfur atom into dethiobiotin via a radical- based mechanism
- Gene Name:
- bioB
- Locus Tag:
- PA0500
- Molecular weight:
- 39.1 kDa
Reactions
| Dethiobiotin + sulfur + 2 S-adenosyl-L-methionine = biotin + 2 L-methionine + 2 5'-deoxyadenosine. |
- General function:
- Involved in methionine synthase activity
- Specific function:
- Catalyzes the transfer of a methyl group from methyl- cobalamin to homocysteine, yielding enzyme-bound cob(I)alamin and methionine. Subsequently, remethylates the cofactor using methyltetrahydrofolate
- Gene Name:
- metH
- Locus Tag:
- PA1843
- Molecular weight:
- 135.1 kDa
Reactions
| 5-methyltetrahydrofolate + L-homocysteine = tetrahydrofolate + L-methionine. |
- General function:
- Involved in 5-methyltetrahydropteroyltriglutamate-homocysteine S-methyltransferase activity
- Specific function:
- Catalyzes the transfer of a methyl group from 5- methyltetrahydrofolate to homocysteine resulting in methionine formation
- Gene Name:
- metE
- Locus Tag:
- PA1927
- Molecular weight:
- 86.2 kDa
Reactions
| 5-methyltetrahydropteroyltri-L-glutamate + L-homocysteine = tetrahydropteroyltri-L-glutamate + L-methionine. |
- General function:
- Involved in coproporphyrinogen oxidase activity
- Specific function:
- Anaerobic transformation of coproporphyrinogen-III into protoporphyrinogen-IX
- Gene Name:
- hemN
- Locus Tag:
- PA1546
- Molecular weight:
- 52.5 kDa
Reactions
| Coproporphyrinogen-III + 2 S-adenosyl-L-methionine = protoporphyrinogen-IX + 2 CO(2) + 2 L-methionine + 2 5'-deoxyadenosine. |
- General function:
- Involved in coproporphyrinogen oxidase activity
- Specific function:
- Not Available
- Gene Name:
- yggW
- Locus Tag:
- PA0386
- Molecular weight:
- 42.5 kDa
- General function:
- Involved in catalytic activity
- Specific function:
- Catalyzes the radical-mediated insertion of two sulfur atoms into the C-6 and C-8 positions of the octanoyl moiety bound to the lipoyl domains of lipoate-dependent enzymes, thereby converting the octanoylated domains into lipoylated derivatives. Free octanoate is not a substrate for lipA
- Gene Name:
- lipA
- Locus Tag:
- PA2862
- Molecular weight:
- 32.7 kDa
Reactions
| Protein N(6)-(octanoyl)lysine + 2 sulfur + 2 S-adenosyl-L-methionine = protein N(6)-(lipoyl)lysine + 2 L-methionine + 2 5'-deoxyadenosine. |
- General function:
- Involved in catalytic activity
- Specific function:
- Catalyzes the rearrangement of 1-deoxy-D-xylulose 5- phosphate (DXP) to produce the thiazole phosphate moiety of thiamine. Sulfur is provided by the thiocarboxylate moiety of the carrier protein ThiS. In vitro, sulfur can be provided by H(2)S
- Gene Name:
- thiG
- Locus Tag:
- PA0381
- Molecular weight:
- 28.2 kDa
Reactions
| 1-deoxy-D-xylulose 5-phosphate + 2-iminoacetate + thiocarboxy-adenylate-[sulfur-carrier protein ThiS] = 2-((2R,5Z)-2-carboxy-4-methylthiazol-5(2H)-ylidene)ethyl phosphate + [sulfur-carrier protein ThiS] + 2 H(2)O. |
- General function:
- Involved in thiamine biosynthetic process
- Specific function:
- Catalyzes the synthesis of the hydroxymethylpyrimidine phosphate (HMP-P) moiety of thiamine from aminoimidazole ribotide (AIR) in a radical S-adenosyl-L-methionine (SAM)-dependent reaction
- Gene Name:
- thiC
- Locus Tag:
- PA4973
- Molecular weight:
- 69.8 kDa
Reactions
| 5-amino-1-(5-phospho-D-ribosyl)imidazole + S-adenosyl-L-methionine = 4-amino-2-methyl-5-phosphomethylpyrimidine + 5'-deoxyadenosine + L-methionine + formate + CO. |
- General function:
- Involved in electron carrier activity
- Specific function:
- Participates in various redox reactions through the reversible oxidation of its active center dithiol to a disulfide and catalyzes dithiol-disulfide exchange reactions
- Gene Name:
- trxA
- Locus Tag:
- PA5240
- Molecular weight:
- 11.9 kDa
- General function:
- Not Available
- Specific function:
- Not Available
- Gene Name:
- yncA
- Locus Tag:
- PA4866
- Molecular weight:
- 18.7 kDa
- General function:
- Translation, ribosomal structure and biogenesis
- Specific function:
- Transfers and isomerizes the ribose moiety from AdoMet to the 7-aminomethyl group of 7-deazaguanine (preQ1-tRNA) to give epoxyqueuosine (oQ-tRNA)
- Gene Name:
- queA
- Locus Tag:
- PA3824
- Molecular weight:
- 38.2 kDa
Reactions
| S-adenosylmethionine + 7-aminomethyl-7-deazaguanosine = methionine + adenine + epoxyqueuosine. |
- General function:
- Involved in 4 iron, 4 sulfur cluster binding
- Specific function:
- Specifically methylates position 2 of adenine 2503 in 23S rRNA
- Gene Name:
- rlmN
- Locus Tag:
- PA3806
- Molecular weight:
- 41.7 kDa
Reactions
| 2 S-adenosyl-L-methionine + adenine(2503) in 23S rRNA = S-adenosyl-L-homocysteine + L-methionine + 5'-deoxyadenosine + 2-methyladenine(2503) in 23S rRNA. |
- General function:
- tRNA methylthiolation
- Specific function:
- Catalyzes the methylthiolation of N6-(dimethylallyl)adenosine (i(6)A), leading to the formation of 2-methylthio-N6-(dimethylallyl)adenosine (ms(2)i(6)A) at position 37 in tRNAs that read codons beginning with uridine.
- Gene Name:
- miaB
- Locus Tag:
- PA3980
- Molecular weight:
- 50 kDa
Reactions
| N(6)-dimethylallyladenine(37) in tRNA + sulfur-(sulfur carrier) + 2 S-adenosyl-L-methionine = 2-methylthio-N(6)-dimethylallyladenine(37) in tRNA + S-adenosyl-L-homocysteine + (sulfur carrier) + L-methionine + 5'-deoxyadenosine |
- General function:
- RNA modification
- Specific function:
- Catalyzes the methylthiolation of the residue Asp-89 of ribosomal protein S12.
- Gene Name:
- rimO
- Locus Tag:
- PA0916
- Molecular weight:
- 49 kDa
Reactions
| L-aspartate-[ribosomal protein S12] + sulfur-(sulfur carrier) + 2 S-adenosyl-L-methionine = 3-methylthio-L-aspartate-[ribosomal protein S12] + S-adenosyl-L-homocysteine + (sulfur carrier) + L-methionine + 5'-deoxyadenosine |
Transporters
- General function:
- Involved in nucleotide binding
- Specific function:
- Probably part of a binding-protein-dependent transport system yecCS for an amino acid. Probably responsible for energy coupling to the transport system
- Gene Name:
- yecC
- Locus Tag:
- PA5152
- Molecular weight:
- 28.4 kDa