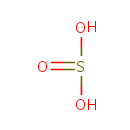
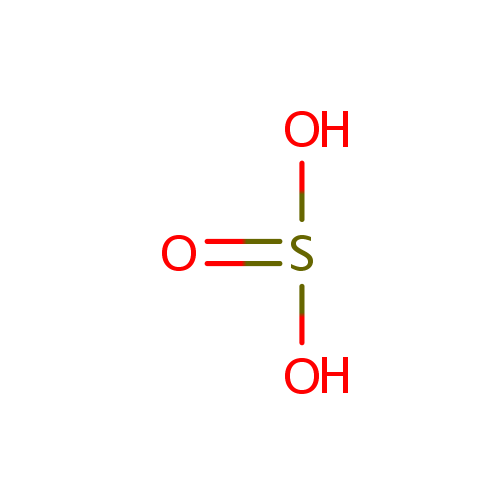
Sulfite (PAMDB000100)
Enzymes
- General function:
- Involved in thiosulfate sulfurtransferase activity
- Specific function:
- Catalyzes, although with low efficiency, the sulfur transfer reaction from thiosulfate to cyanide. The relatively low affinity of glpE for both thiosulfate and cyanide suggests that these compounds are not the physiological substrates. Thioredoxin 1 or related dithiol proteins could instead be the physiological sulfur-acceptor substrate. Possible association with the metabolism of glycerol-phosphate remains to be elucidated
- Gene Name:
- glpE
- Locus Tag:
- PA0589
- Molecular weight:
- 12 kDa
Reactions
| Thiosulfate + cyanide = sulfite + thiocyanate. |
- General function:
- Involved in cysteine biosynthetic process from serine
- Specific function:
- O(3)-acetyl-L-serine + H(2)S = L-cysteine + acetate
- Gene Name:
- cysK
- Locus Tag:
- PA2709
- Molecular weight:
- 34.3 kDa
Reactions
| O(3)-acetyl-L-serine + H(2)S = L-cysteine + acetate. |
| 3-chloro-L-alanine + thioglycolate = S-carboxymethyl-L-cysteine + chloride. |
- General function:
- Involved in cysteine biosynthetic process from serine
- Specific function:
- Two cysteine synthase enzymes are found. Both catalyze the same reaction. Cysteine synthase B can also use thiosulfate in place of sulfide to give cysteine thiosulfonate as a product
- Gene Name:
- cysM
- Locus Tag:
- PA0932
- Molecular weight:
- 32.4 kDa
Reactions
| O(3)-acetyl-L-serine + H(2)S = L-cysteine + acetate. |
- General function:
- Involved in sulfite reductase (NADPH) activity
- Specific function:
- Component of the sulfite reductase complex that catalyzes the 6-electron reduction of sulfite to sulfide. This is one of several activities required for the biosynthesis of L- cysteine from sulfate
- Gene Name:
- cysI
- Locus Tag:
- PA1838
- Molecular weight:
- 62.1 kDa
Reactions
| H(2)S + 3 NADP(+) + 3 H(2)O = sulfite + 3 NADPH. |
- General function:
- Involved in phosphoadenylyl-sulfate reductase (thioredoxin) activity
- Specific function:
- Reduction of activated sulfate into sulfite
- Gene Name:
- cysH
- Locus Tag:
- PA1756
- Molecular weight:
- 30.2 kDa
Reactions
| Adenosine 3',5'-bisphosphate + sulfite + thioredoxin disulfide = 3'-phosphoadenylyl sulfate + thioredoxin. |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Catalyzes the conversion of taurine and alpha ketoglutarate to sulfite, aminoacetaldehyde and succinate. Required for the utilization of taurine (2-aminoethanesulfonic acid) as an alternative sulfur source. Pentane-sulfonic acid, 3- (N-morpholino)propanesulfonic acid and 1,3-dioxo-2- isoindolineethanesulfonic acid are also substrates for this enzyme
- Gene Name:
- tauD
- Locus Tag:
- PA3935
- Molecular weight:
- 31 kDa
Reactions
| Taurine + 2-oxoglutarate + O(2) = sulfite + aminoacetaldehyde + succinate + CO(2). |
- General function:
- Involved in FMN reductase activity
- Specific function:
- Catalyzes an NAD(P)H-dependent reduction of FMN, but is also able to reduce FAD or riboflavin
- Gene Name:
- ssuE
- Locus Tag:
- PA3446
- Molecular weight:
- 21.5 kDa
Reactions
| FMNH(2) + NADP(+) = FMN + NADPH. |
- General function:
- Involved in alkanesulfonate monooxygenase activity
- Specific function:
- Involved in desulfonation of aliphatic sulfonates. Catalyzes the conversion of pentanesulfonic acid to sulfite and pentaldehyde and is able to desulfonate a wide range of sulfonated substrates including C-2 to C-10 unsubstituted linear alkanesulfonates, substituted ethanesulfonic acids and sulfonated buffers
- Gene Name:
- ssuD
- Locus Tag:
- PA3444
- Molecular weight:
- 41.6 kDa
Reactions
| An alkanesufonate (R-CH(2)-SO(3)H) + FMNH(2) + O(2) = an aldehyde (R-CHO) + FMN + sulfite + H(2)O. |
- General function:
- Involved in transporter activity
- Specific function:
- Part of a binding-protein-dependent transport system for aliphatic sulfonates. Probably responsible for the translocation of the substrate across the membrane
- Gene Name:
- ssuC
- Locus Tag:
- PA3443
- Molecular weight:
- 28.5 kDa
- General function:
- Involved in electron carrier activity
- Specific function:
- Monothiol glutaredoxin involved in the biogenesis of iron-sulfur clusters (Probable)
- Gene Name:
- grxD
- Locus Tag:
- PA3533
- Molecular weight:
- 11.8 kDa
- General function:
- Involved in electron carrier activity
- Specific function:
- The disulfide bond functions as an electron carrier in the glutathione-dependent synthesis of deoxyribonucleotides by the enzyme ribonucleotide reductase. In addition, it is also involved in reducing some disulfides in a coupled system with glutathione reductase
- Gene Name:
- grxC
- Locus Tag:
- PA5129
- Molecular weight:
- 9.2 kDa
- General function:
- Involved in electron carrier activity
- Specific function:
- Participates in various redox reactions through the reversible oxidation of its active center dithiol to a disulfide and catalyzes dithiol-disulfide exchange reactions
- Gene Name:
- trxA
- Locus Tag:
- PA5240
- Molecular weight:
- 11.9 kDa
Transporters
- General function:
- Involved in transporter activity
- Specific function:
- Part of a binding-protein-dependent transport system for aliphatic sulfonates. Probably responsible for the translocation of the substrate across the membrane
- Gene Name:
- ssuC
- Locus Tag:
- PA3443
- Molecular weight:
- 28.5 kDa