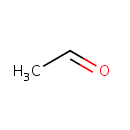
Acetaldehyde (PAMDB000217)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000217 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | Acetaldehyde | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | Acetaldehyde is a colorless, flammable liquid used in the manufacture of acetic acid, perfumes, and flavors. It is also an intermediate in the metabolism of alcohol. Small amounts of acetaldehyde are produced naturally through gut microbial fermentation. Acetaldehyde is produced through the action of alcohol dehydrogenase on ethanol and is somewhate more toxic than ethanol. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C2H4O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 44.0526 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 44.02621475 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | IKHGUXGNUITLKF-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C2H4O/c1-2-3/h2H,1H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 75-07-0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | acetaldehyde | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | acetaldehyde | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | CC=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as short-chain aldehydes. These are an aldehyde with a chain length containing between 2 and 5 carbon atoms. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organooxygen compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carbonyl compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Aldehydes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Short-chain aldehydes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Liquid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | -123 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | Acetaldehyde + Coenzyme A + NAD <> Acetyl-CoA + Hydrogen ion + NADH Ethanol + NAD <> Acetaldehyde + Hydrogen ion + NADH Ethanolamine > Acetaldehyde + Ammonium L-Threonine <> Acetaldehyde + Glycine L-Allothreonine > Acetaldehyde + Glycine 4-Hydroxy-2-oxopentanoate > Acetaldehyde + Pyruvic acid Ethanesulfonate + FMNH + Oxygen > Acetaldehyde + Flavin Mononucleotide + Hydrogen ion + Water + Sulfite Acetaldehyde + Water + NAD > Acetic acid +2 Hydrogen ion + NADH Dihydroneopterin triphosphate + Water > Acetaldehyde + 6-Carboxy-5,6,7,8-tetrahydropterin + Hydrogen ion + Triphosphate Acetaldehyde + Water + NADP > Acetic acid +2 Hydrogen ion + NADPH Ethanolamine <> Acetaldehyde + Ammonia Hydrogen ion + Pyruvic acid + Acetaldehyde > acetoin + Carbon dioxide Deoxyribose 5-phosphate <> Acetaldehyde + D-Glyceraldehyde 3-phosphate Ethanolamine > Hydrogen ion + Acetaldehyde + Ammonia DL-allothreonine <> Acetaldehyde + Glycine More...1-Ethyladenine + Oxygen + Oxoglutaric acid > Adenine + Carbon dioxide + Acetaldehyde + Succinic acid Dihydroneopterin triphosphate + Water > 6-Carboxy-5,6,7,8-tetrahydropterin + Acetaldehyde + Triphosphate 4-hydroxy-2-oxopentanoate + 4-Hydroxy-2-oxopentanoate > Pyruvic acid + Acetaldehyde 7,8-dihydroneopterin 3'-triphosphate + Water > Acetaldehyde + Triphosphate +2 Hydrogen ion + 6-Carboxy-5,6,7,8-tetrahydropterin + Triphosphate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Wertheim, E. Laboratory preparation of acetaldehyde. Journal of the American Chemical Society (1922), 44 2658-9. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in catalytic activity
- Specific function:
- Interconversion of serine and glycine
- Gene Name:
- glyA
- Locus Tag:
- PA4602
- Molecular weight:
- 45.2 kDa
Reactions
| 5,10-methylenetetrahydrofolate + glycine + H(2)O = tetrahydrofolate + L-serine. |
- General function:
- Involved in catalytic activity
- Specific function:
- Ethanolamine = acetaldehyde + NH(3)
- Gene Name:
- eutB
- Locus Tag:
- PA4024
- Molecular weight:
- 50.2 kDa
Reactions
| Ethanolamine = acetaldehyde + NH(3). |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Involved in the breakdown of putrescine. Was previously shown to have a weak but measurable ALDH enzyme activity that prefers NADP over NAD as coenzyme
- Gene Name:
- puuC
- Locus Tag:
- PA5312
- Molecular weight:
- 53.1 kDa
Reactions
| Gamma-glutamyl-gamma-aminobutyraldehyde + NAD(+) + H(2)O = gamma-glutamyl-gamma-aminobutyrate + NADH. |
| An aldehyde + NAD(P)(+) + H(2)O = a carboxylate + NAD(P)H. |
- General function:
- Involved in zinc ion binding
- Specific function:
- Has high formaldehyde dehydrogenase activity in the presence of glutathione and catalyzes the oxidation of normal alcohols in a reaction that is not GSH-dependent. In addition, hemithiolacetals other than those formed from GSH, including omega-thiol fatty acids, also are substrates
- Gene Name:
- frmA
- Locus Tag:
- PA3629
- Molecular weight:
- 39.2 kDa
Reactions
| S-(hydroxymethyl)glutathione + NAD(P)(+) = S-formylglutathione + NAD(P)H. |
| An alcohol + NAD(+) = an aldehyde or ketone + NADH. |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Catalyzes the NADP-dependent oxidation of diverse aldehydes such as chloroacetaldehyde, acetaldehyde, propionaldehyde, benzaldehyde, mafosfamide, 4- hydroperoxycyclophosphamide. Its preferred substrates are acetaldehyde and chloroacetaldehyde
- Gene Name:
- aldB
- Locus Tag:
- PA1984
- Molecular weight:
- 54.9 kDa
- General function:
- Coenzyme transport and metabolism
- Specific function:
- Catalyzes the conversion of 7,8-dihydroneopterin triphosphate (H2NTP) to 6-carboxy-5,6,7,8-tetrahydropterin (CPH4) and acetaldehyde. Can also convert 6-pyruvoyltetrahydropterin (PPH4) and sepiapterin to CPH4; these 2 compounds are probably intermediates in the reaction from H2NTP
- Gene Name:
- queD
- Locus Tag:
- PA2666
- Molecular weight:
- 13.8 kDa
Reactions
| 7,8-dihydroneopterin 3'-triphosphate + H(2)O = 6-carboxy-5,6,7,8-tetrahydropterin + acetaldehyde + triphosphate. |
- General function:
- Involved in lyase activity
- Specific function:
- Catalyzes the cleavage of L-allo-threonine and L- threonine to glycine and acetaldehyde. L-threo-phenylserine and L- erythro-phenylserine are also good substrates
- Gene Name:
- ltaE
- Locus Tag:
- PA0902
- Molecular weight:
- 35.4 kDa
Reactions
| L-threonine = glycine + acetaldehyde. |
| L-allo-threonine = glycine + acetaldehyde. |
- General function:
- Involved in alkanesulfonate monooxygenase activity
- Specific function:
- Involved in desulfonation of aliphatic sulfonates. Catalyzes the conversion of pentanesulfonic acid to sulfite and pentaldehyde and is able to desulfonate a wide range of sulfonated substrates including C-2 to C-10 unsubstituted linear alkanesulfonates, substituted ethanesulfonic acids and sulfonated buffers
- Gene Name:
- ssuD
- Locus Tag:
- PA3444
- Molecular weight:
- 41.6 kDa
Reactions
| An alkanesufonate (R-CH(2)-SO(3)H) + FMNH(2) + O(2) = an aldehyde (R-CHO) + FMN + sulfite + H(2)O. |