|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB001656 |
|---|
|
Identification |
|---|
| Name: |
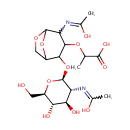
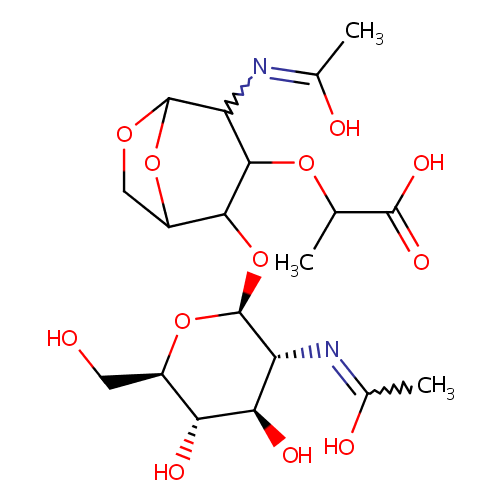
N-Acetyl-D-glucosamine(anhydrous)N-Acetylmuramic acid |
|---|
| Description: | N-acetyl-d-glucosamine(anhydrous)n-acetylmuramic acid belongs to the class of Hexoses. These are monosaccharides in which the sugar unit is a hexose. (inferred from compound structure) |
|---|
|
Structure |
|
|---|
| Synonyms: | - N-Acetyl-D-glucosamine(anhydrous)N-Acetylmuramate
|
|---|
|
Chemical Formula: |
C19H30N2O12 |
|---|
| Average Molecular Weight: |
478.4477 |
|---|
| Monoisotopic Molecular
Weight: |
478.179874434 |
|---|
| InChI Key: |
MWWQKONGFKUAEK-AJSMYUJSSA-N |
|---|
| InChI: | InChI=1S/C19H30N2O12/c1-6(17(27)28)30-16-12(21-8(3)24)18-29-5-10(32-18)15(16)33-19-11(20-7(2)23)14(26)13(25)9(4-22)31-19/h6,9-16,18-19,22,25-26H,4-5H2,1-3H3,(H,20,23)(H,21,24)(H,27,28)/t6?,9-,10?,11-,12?,13-,14-,15?,16?,18?,19+/m1/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | 2-[(2-{[(2S,3R,4R,5S,6R)-4,5-dihydroxy-3-[(1-hydroxyethylidene)amino]-6-(hydroxymethyl)oxan-2-yl]oxy}-4-[(1-hydroxyethylidene)amino]-6,8-dioxabicyclo[3.2.1]octan-3-yl)oxy]propanoic acid |
|---|
|
Traditional IUPAC Name: |
2-[(2-{[(2S,3R,4R,5S,6R)-4,5-dihydroxy-3-[(1-hydroxyethylidene)amino]-6-(hydroxymethyl)oxan-2-yl]oxy}-4-[(1-hydroxyethylidene)amino]-6,8-dioxabicyclo[3.2.1]octan-3-yl)oxy]propanoic acid |
|---|
| SMILES: | CC(OC1C(O[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2N=C(C)O)C2COC(O2)C1N=C(C)O)C(O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as n-acyl-alpha-hexosamines. These are carbohydrate derivatives containing a hexose moiety in which the oxygen atom is replaced by an n-acyl group. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Organooxygen compounds |
|---|
|
Class |
Carbohydrates and carbohydrate conjugates |
|---|
| Sub Class | Aminosaccharides |
|---|
|
Direct Parent |
N-acyl-alpha-hexosamines |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- N-acyl-alpha-hexosamine
- Glucosamine
- Amino sugar
- O-glycosyl compound
- Glycosyl compound
- Disaccharide
- Oxepane
- Oxane
- Meta-dioxolane
- Secondary alcohol
- 1,2-diol
- Oxacycle
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Monocarboxylic acid or derivatives
- Ether
- Dialkyl ether
- Carboxylic acid
- Carboxylic acid derivative
- Carboximidic acid derivative
- Carboximidic acid
- Acetal
- Hydrocarbon derivative
- Primary alcohol
- Organonitrogen compound
- Carbonyl group
- Alcohol
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework |
Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
Not Available |
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | 45479497 | | Kegg ID | Not Available | | ChemSpider ID | Not Available | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|