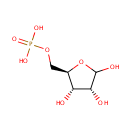
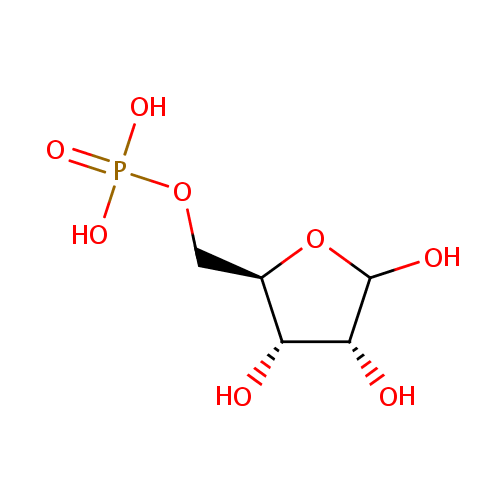
D-Ribose-5-phosphate (PAMDB000431)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000431 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | D-Ribose-5-phosphate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | D-ribose-5-phosphate is a member of the chemical class known as Pentoses. These are monosaccharides in which the carbohydrate moiety contains five carbon atoms. Ribose 5-phosphate is both a product and an intermediate of the pentose phosphate pathway. The last step of the oxidative reactions in the pentose phosphate pathway is the production of ribulose-5-phosphate. Ribulose-5-phosphate can reversibly isomerize to ribose-5-phosphate. Ribulose-5-phosphate can alternatively undergo a series of isomerizations as well as transaldolations and transketolations that result in the production of other pentoses phosphates as well as fructose 6-phosphate and glyceraldehyde-3-phosphate (both intermediates in glycolysis). The enzyme ribose-phosphate diphosphokinase converts ribose-5-phosphate into phosphoribosyl pyrophosphate. (WikiPedia) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C5H11O8P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 230.1098 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 230.01915384 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | KTVPXOYAKDPRHY-SOOFDHNKSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C5H11O8P/c6-3-2(1-12-14(9,10)11)13-5(8)4(3)7/h2-8H,1H2,(H2,9,10,11)/t2-,3-,4-,5?/m1/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 4151-19-3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | {[(2R,3S,4R)-3,4,5-trihydroxyoxolan-2-yl]methoxy}phosphonic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | D-ribofuranose 5-phosphate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | OC1O[C@H](COP(O)(O)=O)[C@@H](O)[C@H]1O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as monosaccharide phosphates. These are monosaccharides comprising a phosphated group linked to the carbohydrate unit. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organooxygen compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carbohydrates and carbohydrate conjugates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Monosaccharides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Monosaccharide phosphates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic heteromonocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | D-Ribose-5-phosphate <> D-Ribulose 5-phosphate D-Ribose-5-phosphate + Xylulose 5-phosphate <> D-Glyceraldehyde 3-phosphate + D-Sedoheptulose 7-phosphate Ribose-1-phosphate <> D-Ribose-5-phosphate Adenosine diphosphate ribose + Water <> Adenosine monophosphate +2 Hydrogen ion + D-Ribose-5-phosphate Water + D-Ribose-5-phosphate > Phosphate + Ribose Adenosine monophosphate + Water <> Adenine + D-Ribose-5-phosphate Adenosine triphosphate + Ribose <> ADP + Hydrogen ion + D-Ribose-5-phosphate Adenosine triphosphate + D-Ribose-5-phosphate <> Adenosine monophosphate + Phosphoribosyl pyrophosphate Adenosine triphosphate + Ribose <> ADP + D-Ribose-5-phosphate Adenosine diphosphate ribose + Water <> Adenosine monophosphate + D-Ribose-5-phosphate D-Ribose-5-phosphate <> D-Ribulose 5-phosphate alpha-D-Ribose 1-phosphate + Ribose-1-phosphate <> D-Ribose-5-phosphate Sedoheptulose 7-phosphate + D-Glyceraldehyde 3-phosphate + D-Sedoheptulose 7-phosphate <> D-Ribose-5-phosphate + Xylulose 5-phosphate Aminofructose 6-phosphate + D-Ribose-5-phosphate <> Iminoerythrose 4-phosphate + Sedoheptulose 7-phosphate Water + Nicotinamide ribotide <> Hydrogen ion + D-Ribose-5-phosphate + Niacinamide Adenosine triphosphate + D-Ribose-5-phosphate <> Hydrogen ion + Phosphoribosyl pyrophosphate + Adenosine monophosphate D-ribose + Adenosine triphosphate > Hydrogen ion + D-Ribose-5-phosphate + ADP D-Ribose-5-phosphate + Uracil <> Water + Pseudouridine 5'-phosphate More...Xylulose 5-phosphate + D-Ribose-5-phosphate + Xylulose 5-phosphate <> D-Sedoheptulose 7-phosphate + D-Glyceraldehyde 3-phosphate + D-Sedoheptulose 7-phosphate + D-Glyceraldehyde 3-phosphate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in magnesium ion binding
- Specific function:
- ATP + D-ribose 5-phosphate = AMP + 5-phospho- alpha-D-ribose 1-diphosphate
- Gene Name:
- prs
- Locus Tag:
- PA4670
- Molecular weight:
- 34 kDa
Reactions
| ATP + D-ribose 5-phosphate = AMP + 5-phospho-alpha-D-ribose 1-diphosphate. |
- General function:
- Involved in ribose-5-phosphate isomerase activity
- Specific function:
- D-ribose 5-phosphate = D-ribulose 5-phosphate
- Gene Name:
- rpiA
- Locus Tag:
- PA0330
- Molecular weight:
- 23.7 kDa
Reactions
| D-ribose 5-phosphate = D-ribulose 5-phosphate. |
- General function:
- Involved in phosphotransferase activity, alcohol group as acceptor
- Specific function:
- ATP + D-ribose = ADP + D-ribose 5-phosphate
- Gene Name:
- rbsK
- Locus Tag:
- PA1950
- Molecular weight:
- 31.8 kDa
Reactions
| ATP + D-ribose = ADP + D-ribose 5-phosphate. |
- General function:
- Involved in catalytic activity
- Specific function:
- Involved in regulation of AMP concentrations
- Gene Name:
- amn
- Locus Tag:
- PA3970
- Molecular weight:
- 56.2 kDa
Reactions
| AMP + H(2)O = D-ribose 5-phosphate + adenine. |
- General function:
- Involved in acid phosphatase activity
- Specific function:
- Dephosphorylates several organic phosphomonoesters and catalyzes the transfer of low-energy phosphate groups from phosphomonoesters to hydroxyl groups of various organic compounds. Preferentially acts on aryl phosphoesters. Might function as a broad-spectrum dephosphorylating enzyme able to scavenge both 3'- and 5'-nucleotides and also additional organic phosphomonoesters
- Gene Name:
- aphA
- Locus Tag:
- PA1409
- Molecular weight:
- 38 kDa
Reactions
| A phosphate monoester + H(2)O = an alcohol + phosphate. |
- General function:
- Involved in catalytic activity
- Specific function:
- Sedoheptulose 7-phosphate + D-glyceraldehyde 3-phosphate = D-ribose 5-phosphate + D-xylulose 5-phosphate
- Gene Name:
- tktA
- Locus Tag:
- PA0548
- Molecular weight:
- 72.2 kDa
Reactions
| Sedoheptulose 7-phosphate + D-glyceraldehyde 3-phosphate = D-ribose 5-phosphate + D-xylulose 5-phosphate. |
- General function:
- Involved in intramolecular transferase activity, phosphotransferases
- Specific function:
- This enzyme participates in both the breakdown and synthesis of glucose
- Gene Name:
- pgm
- Locus Tag:
- PA5131
- Molecular weight:
- 55.6 kDa
Reactions
| Alpha-D-glucose 1-phosphate = alpha-D-glucose 6-phosphate. |
- General function:
- Involved in hydrolase activity, acting on acid anhydrides, in phosphorus-containing anhydrides
- Specific function:
- Acts on ADP-mannose and ADP-glucose as well as ADP- ribose. Prevents glycogen biosynthesis. The reaction catalyzed by this enzyme is a limiting step of the gluconeogenic process
- Gene Name:
- nudF
- Locus Tag:
- PA4971
- Molecular weight:
- 23.1 kDa
Reactions
| ADP-ribose + H(2)O = AMP + D-ribose 5-phosphate. |