L-Proline (PAMDB000064)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000064 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
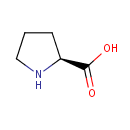
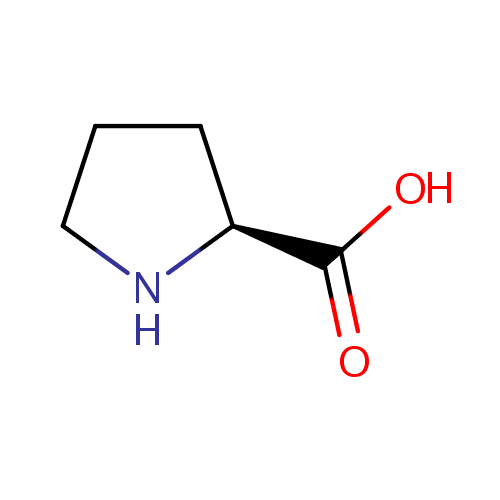
| Name: | L-Proline | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | L-Proline is one of the twenty amino acids used in living organisms as the building blocks of proteins. Proline is sometimes called an imino acid, although the IUPAC definition of an imine requires a carbon-nitrogen double bond. Proline is a non-essential amino acid that is synthesized from glutamic acid. It is an essential component of collagen and is important for proper functioning of joints and tendons. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C5H9NO2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 115.1305 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 115.063328537 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | ONIBWKKTOPOVIA-BYPYZUCNSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C5H9NO2/c7-5(8)4-2-1-3-6-4/h4,6H,1-3H2,(H,7,8)/t4-/m0/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 147-85-3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | (2S)-pyrrolidine-2-carboxylic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | L-proline | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | OC(=O)[C@@H]1CCCN1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as l-alpha-amino acids. These are alpha amino acids which have the L-configuration of the alpha-carbon atom. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carboxylic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Amino acids, peptides, and analogues | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | L-alpha-amino acids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic heteromonocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | 221 °C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | Adenosine triphosphate + Water + L-Proline > ADP + Hydrogen ion + Phosphate + L-Proline Adenosine triphosphate + Water + L-Proline > ADP + Hydrogen ion + Phosphate + L-Proline Water + L-Prolinylglycine > Glycine + L-Proline Adenosine triphosphate + L-Proline + tRNA(Pro) + tRNA(Pro) <> Adenosine monophosphate + Pyrophosphate + L-Prolyl-tRNA(Pro) + L-Prolyl-tRNA(Pro) L-D-1-Pyrroline-5-carboxylic acid + 2 Hydrogen ion + NADPH > NADP + L-Proline FAD + L-Proline > L-D-1-Pyrroline-5-carboxylic acid + FADH2 + Hydrogen ion L-Proline + NAD <> L-D-1-Pyrroline-5-carboxylic acid + NADH + Hydrogen ion L-Proline + Acceptor + Quinone <> L-D-1-Pyrroline-5-carboxylic acid + Reduced acceptor + (S)-1-pyrroline-5-carboxylate + Hydroquinone Adenosine triphosphate + L-Proline + tRNA(Pro) <> Adenosine monophosphate + Pyrophosphate + L-Prolyl-tRNA(Pro) a dipeptide with proline at carboxy terminal + Water L-Proline + a standard α amino acid -->-->NAD(P)<sup>+</sup> + L-Proline < NAD(P)H + L-D-1-Pyrroline-5-carboxylic acid + Hydrogen ion L-Proline + an oxidized electron acceptor > L-D-1-Pyrroline-5-carboxylic acid + a reduced electron acceptor + Hydrogen ion glycylproline + Water > Glycine + L-Proline L-Proline + NAD(P)(+) > L-D-1-Pyrroline-5-carboxylic acid + NAD(P)H L-Proline + acceptor > (S)-1-pyrroline-5-carboxylate + reduced acceptor L-Proline + NAD + NADP <> (S)-1-pyrroline-5-carboxylate + NADH + NADPH + Hydrogen ion 1-Pyrroline-5-carboxylic acid + Hydrogen ion + NADPH + L-D-1-Pyrroline-5-carboxylic acid + NADPH > NADP + L-Proline + L-Proline L-Proline + Ubiquinone-1 + L-Proline > Hydrogen ion + Ubiquinol-1 + 1-Pyrroline-5-carboxylic acid + L-D-1-Pyrroline-5-carboxylic acid L-Proline + Adenosine triphosphate + Hydrogen ion + tRNA(Pro) + L-Proline > Adenosine monophosphate + Pyrophosphate + L-prolyl-tRNA(Pro) L-Proline + Adenosine triphosphate + Water + L-Proline > L-Proline + Adenosine diphosphate + Phosphate + Hydrogen ion + ADP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Itoh, Tamio. Synthesis of L-proline from L-glutamine. Bulletin of the Chemical Society of Japan (1963), 36(1), 25-9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in proteolysis
- Specific function:
- Aminopeptidase N is involved in the degradation of intracellular peptides generated by protein breakdown during normal growth as well as in response to nutrient starvation
- Gene Name:
- pepN
- Locus Tag:
- PA3083
- Molecular weight:
- 100 kDa
Reactions
| Release of an N-terminal amino acid, Xaa-|-Yaa- from a peptide, amide or arylamide. Xaa is preferably Ala, but may be most amino acids including Pro (slow action). When a terminal hydrophobic residue is followed by a prolyl residue, the two may be released as an intact Xaa-Pro dipeptide. |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Oxidizes proline to glutamate for use as a carbon and nitrogen source and also function as a transcriptional repressor of the put operon
- Gene Name:
- putA
- Locus Tag:
- PA0782
- Molecular weight:
- 115.6 kDa
Reactions
| L-proline + acceptor = (S)-1-pyrroline-5-carboxylate + reduced acceptor. |
| (S)-1-pyrroline-5-carboxylate + NAD(P)(+) + 2 H(2)O = L-glutamate + NAD(P)H. |
- General function:
- Involved in oxidation-reduction process
- Specific function:
- L-proline + NAD(P)(+) = 1-pyrroline-5- carboxylate + NAD(P)H
- Gene Name:
- proC
- Locus Tag:
- PA0393
- Molecular weight:
- 28.1 kDa
Reactions
| L-proline + NAD(P)(+) = 1-pyrroline-5-carboxylate + NAD(P)H. |
- General function:
- Involved in nucleotide binding
- Specific function:
- Involved in a multicomponent binding-protein-dependent transport system for glycine betaine/L-proline
- Gene Name:
- proV
- Locus Tag:
- PA5094
- Molecular weight:
- 30.7 kDa
- General function:
- Involved in nucleotide binding
- Specific function:
- Catalyzes the attachment of proline to tRNA(Pro) in a two-step reaction:proline is first activated by ATP to form Pro- AMP and then transferred to the acceptor end of tRNA(Pro). As ProRS can inadvertently accommodate and process non-cognate amino acids such as alanine and cysteine, to avoid such errors it has two additional distinct editing activities against alanine. One activity is designated as 'pretransfer' editing and involves the tRNA(Pro)-independent hydrolysis of activated Ala-AMP. The other activity is designated 'posttransfer' editing and involves deacylation of mischarged Ala-tRNA(Pro). Misacylated Cys-tRNA(Pro) is not edited by ProRS, but instead may be edited in trans by ybaK
- Gene Name:
- proS
- Locus Tag:
- PA0956
- Molecular weight:
- 63.1 kDa
Reactions
| ATP + L-proline + tRNA(Pro) = AMP + diphosphate + L-prolyl-tRNA(Pro). |
- General function:
- Involved in aminopeptidase activity
- Specific function:
- Presumably involved in the processing and regular turnover of intracellular proteins. Catalyzes the removal of unsubstituted N-terminal amino acids from various peptides. Required for plasmid ColE1 site-specific recombination but not in its aminopeptidase activity. Could act as a structural component of the putative nucleoprotein complex in which the Xer recombination reaction takes place
- Gene Name:
- pepA
- Locus Tag:
- PA3831
- Molecular weight:
- 52.3 kDa
Reactions
| Release of an N-terminal amino acid, Xaa-|-Yaa-, in which Xaa is preferably Leu, but may be other amino acids including Pro although not Arg or Lys, and Yaa may be Pro. Amino acid amides and methyl esters are also readily hydrolyzed, but rates on arylamides are exceedingly low. |
| Release of an N-terminal amino acid, preferentially leucine, but not glutamic or aspartic acids. |
Transporters
- General function:
- Involved in proteolysis
- Specific function:
- Aminopeptidase N is involved in the degradation of intracellular peptides generated by protein breakdown during normal growth as well as in response to nutrient starvation
- Gene Name:
- pepN
- Locus Tag:
- PA3083
- Molecular weight:
- 100 kDa
Reactions
| Release of an N-terminal amino acid, Xaa-|-Yaa- from a peptide, amide or arylamide. Xaa is preferably Ala, but may be most amino acids including Pro (slow action). When a terminal hydrophobic residue is followed by a prolyl residue, the two may be released as an intact Xaa-Pro dipeptide. |
- General function:
- Involved in proline:sodium symporter activity
- Specific function:
- Catalyzes the sodium-dependent uptake of extracellular L-proline. This protein is also capable of using lithium as the transport cation. Also catalyzes the uptake of propionate
- Gene Name:
- putP
- Locus Tag:
- PA0783
- Molecular weight:
- 54.4 kDa
- General function:
- Involved in nucleotide binding
- Specific function:
- Probably part of a binding-protein-dependent transport system yecCS for an amino acid. Probably responsible for energy coupling to the transport system
- Gene Name:
- yecC
- Locus Tag:
- PA5152
- Molecular weight:
- 28.4 kDa
- General function:
- Involved in transport
- Specific function:
- Permease that is involved in the transport across the cytoplasmic membrane of proline
- Gene Name:
- proY
- Locus Tag:
- PA5097
- Molecular weight:
- 50.2 kDa