|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB120592 |
|---|
|
Identification |
|---|
| Name: |
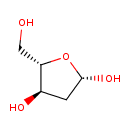
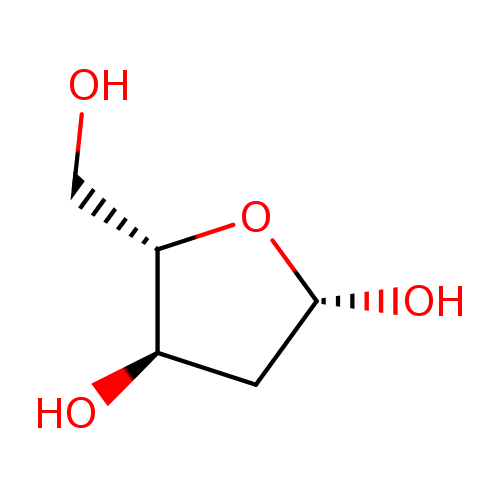
2'-deoxyribose |
|---|
| Description: | A deoxypentose that is D-ribose in which the hydroxy group at position C-2 is replaced by hydrogen. |
|---|
|
Structure |
|
|---|
| Synonyms: | - 2-Deoxy-D-erythro-pentose
- 2-Deoxy-D-ribose
- 2-deoxy-D-ribose
- D-dRib
- Deoxyribose
- Thyminose
|
|---|
|
Chemical Formula: |
C5H10O4 |
|---|
| Average Molecular Weight: |
134.132 |
|---|
| Monoisotopic Molecular
Weight: |
134.0579 |
|---|
| InChI Key: |
ASJSAQIRZKANQN-UHFFFAOYSA-N |
|---|
| InChI: | InChI=1S/C5H10O4/c6-2-1-4(8)5(9)3-7/h2,4-5,7-9H,1,3H2 |
|---|
| CAS
number: |
533-67-5 |
|---|
| IUPAC Name: | 2-deoxy-D-erythro-pentose |
|---|
|
Traditional IUPAC Name: |
2-deoxyribose |
|---|
| SMILES: | C(O)C(O)C(O)CC=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as pentoses. These are monosaccharides in which the carbohydrate moiety contains five carbon atoms. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Organic oxygen compounds |
|---|
| Sub Class | Organooxygen compounds |
|---|
|
Direct Parent |
Pentoses |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Pentose monosaccharide
- Oxolane
- Secondary alcohol
- Hemiacetal
- Oxacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Primary alcohol
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | 0 |
|---|
|
Melting point: |
91 °C |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | 91 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Roux A, Xu Y, Heilier JF, Olivier MF, Ezan E, Tabet JC, Junot C (2012)Annotation of the human adult urinary metabolome and metabolite identification using ultra high performance liquid chromatography coupled to a linear quadrupole ion trap-Orbitrap mass spectrometer. Analytical chemistry 84, Pubmed: 22770225
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Download (PDF) |
|---|
|
Links |
|---|
| External Links: |
|
|---|