|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB003410 |
|---|
|
Identification |
|---|
| Name: |
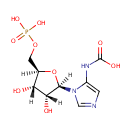
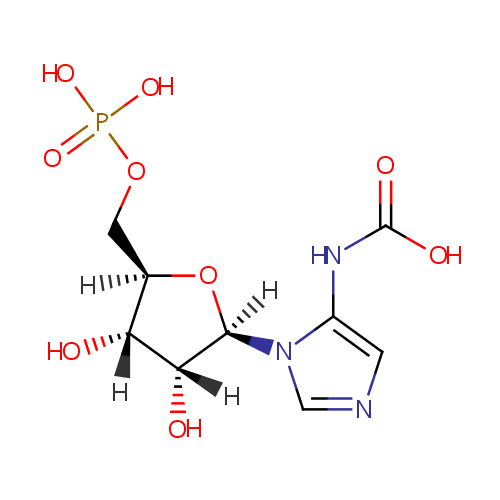
5-carboxyamino-1-(5-phospho-D-ribosyl)imidazole |
|---|
| Description: | A 1-(phosphoribosyl)imidazole having the phospho group at the 5'-position and a carboxyamino group at the 5-position on the imidazole ring |
|---|
|
Structure |
|
|---|
| Synonyms: | Not Available |
|---|
|
Chemical Formula: |
C9H14N3O9P |
|---|
| Average Molecular Weight: |
339.1959 |
|---|
| Monoisotopic Molecular
Weight: |
339.046765573 |
|---|
| InChI Key: |
JHLXDWGVSYMXPL-XVFCMESISA-N |
|---|
| InChI: | InChI=1S/C9H14N3O9P/c13-6-4(2-20-22(17,18)19)21-8(7(6)14)12-3-10-1-5(12)11-9(15)16/h1,3-4,6-8,11,13-14H,2H2,(H,15,16)(H2,17,18,19)/t4-,6-,7-,8-/m1/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | {1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]-1H-imidazol-5-yl}carbamic acid |
|---|
|
Traditional IUPAC Name: |
3-[(2R,3R,4S,5R)-3,4-dihydroxy-5-[(phosphonooxy)methyl]oxolan-2-yl]imidazol-4-ylcarbamic acid |
|---|
| SMILES: | [H][C@]1(COP(O)(O)=O)O[C@@]([H])(N2C=NC=C2NC(O)=O)[C@]([H])(O)[C@]1([H])O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as 1-phosphoribosyl-imidazoles. These are organic compounds containing the imidazole ring linked to a ribose phosphate through a 1-2 bond. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
|
Class |
Imidazole ribonucleosides and ribonucleotides |
|---|
| Sub Class | 1-phosphoribosyl-imidazoles |
|---|
|
Direct Parent |
1-phosphoribosyl-imidazoles |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- 1-phosphoribosyl-imidazole
- N-glycosyl compound
- Glycosyl compound
- Monosaccharide phosphate
- Monoalkyl phosphate
- Alkyl phosphate
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Organic phosphate
- N-substituted imidazole
- Monosaccharide
- Saccharide
- Aminoimidazole
- Heteroaromatic compound
- Oxolane
- Imidazole
- Azole
- Secondary alcohol
- Carbamic acid derivative
- Carbamic acid
- 1,2-diol
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Monocarboxylic acid or derivatives
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Alcohol
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aromatic heteromonocyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -3 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | 48000 | | HMDB ID | Not Available | | Pubchem Compound ID | 23657851 | | Kegg ID | C15667 | | ChemSpider ID | 21865490 | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|