|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB001637 |
|---|
|
Identification |
|---|
| Name: |
Hexanoate (N-C6:0) |
|---|
| Description: | Hexanoate (n-c6:0) belongs to the class of Carboxylic Acid Salts. These are ionic derivatives of carboxylic acid. (inferred from compound structure)Hexanoic acid (caproic acid), is the carboxylic acid derived from hexane with the general formula C5H11COOH. It is a colorless oily liquid with an odor that is fatty, cheesy, waxy, and like that of goats or other barnyard animals. It is a fatty acid found naturally in various animal fats and oils, and is one of the chemicals that give the decomposing fleshy seed coat of the ginkgo its characteristic unpleasant odor. The primary use of hexanoic acid is in the manufacture of its esters for artificial flavors, and in the manufacture of hexyl derivatives such as hexylphenols. The salts and esters of this acid are known as hexanoates or caproates. Two other acids are named after goats: caprylic (C8) and capric (C10). Along with hexanoic acid, these total 15% in goat milk fat. Caproic, caprylic, and capric acids (capric is a crystal- or wax-like substance, whereas the other 2 are mobile liquids) are not only used for the formation of esters but also commonly used neat in: butter, milk, cream, strawberry, bread, beer, nut, and other flavors. (WikiPedia) |
|---|
|
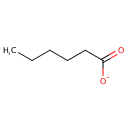
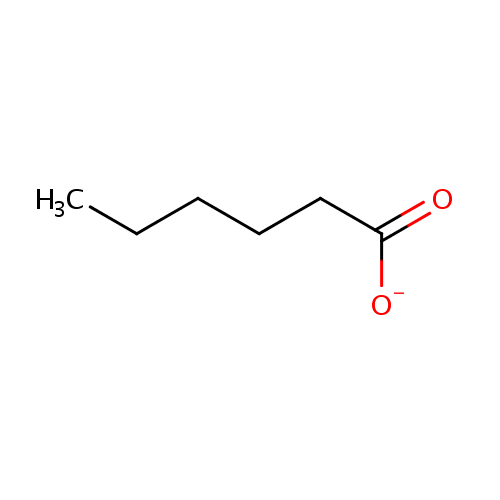
Structure |
|
|---|
| Synonyms: | - Caproate
- Caproic acid
- Hexanoate
- Hexanoate (N-C6:0)
- Hexanoic acid
- Hexanoic acid (N-C6:0)
|
|---|
|
Chemical Formula: |
C6H11O2 |
|---|
| Average Molecular Weight: |
115.1503 |
|---|
| Monoisotopic Molecular
Weight: |
115.075904596 |
|---|
| InChI Key: |
FUZZWVXGSFPDMH-UHFFFAOYSA-M |
|---|
| InChI: | InChI=1S/C6H12O2/c1-2-3-4-5-6(7)8/h2-5H2,1H3,(H,7,8)/p-1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | hexanoate |
|---|
|
Traditional IUPAC Name: |
hexanoate |
|---|
| SMILES: | CCCCCC([O-])=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as medium-chain fatty acids. These are fatty acids with an aliphatic tail that contains between 4 and 12 carbon atoms. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Fatty Acyls |
|---|
| Sub Class | Fatty acids and conjugates |
|---|
|
Direct Parent |
Medium-chain fatty acids |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Medium-chain fatty acid
- Straight chain fatty acid
- Carboxylic acid salt
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Organic anion
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
Not Available |
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Yurtsever D. (2007). Fatty acid methyl ester profiling of Enterococcus and Esherichia coli for microbial source tracking. M.sc. Thesis. Villanova University: U.S.A
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|