|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB001551 |
|---|
|
Identification |
|---|
| Name: |
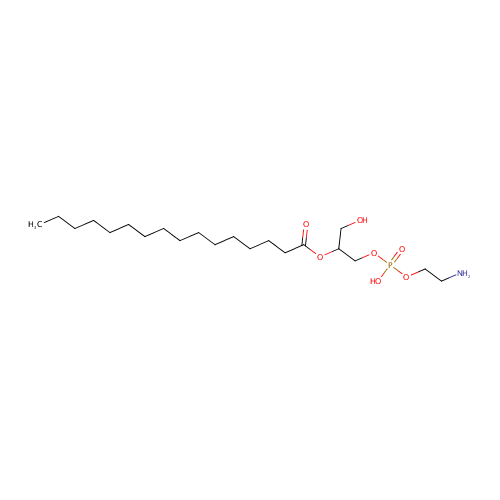
2-Acyl-sn-glycero-3-phosphoethanolamine (N-C16:0) |
|---|
| Description: | 2-acyl-sn-glycero-3-phosphoethanolamine (n-c16:0) belongs to the class of Lysophosphatidylethanolamines. These are glycerophosphoetahnolamines (molecules containing an ethanolamine moiety attached to the phosphate group linked to a glycerol) with one saturated fatty acid bonded to the glycerol moiety through an ester linkage. (inferred from compound structure) |
|---|
|
Structure |
|
|---|
| Synonyms: | - 1-2-aminoethoxy(hydroxy)phosphoryloxy-3-hydroxypropan-2-yl hexadecanoate
- 1-2-Aminoethoxy(hydroxy)phosphoryloxy-3-hydroxypropan-2-yl hexadecanoic acid
|
|---|
|
Chemical Formula: |
C21H44NO7P |
|---|
| Average Molecular Weight: |
453.5503 |
|---|
| Monoisotopic Molecular
Weight: |
453.285539279 |
|---|
| InChI Key: |
CKPBBEOJHAPPBT-UHFFFAOYSA-N |
|---|
| InChI: | InChI=1S/C21H44NO7P/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-21(24)29-20(18-23)19-28-30(25,26)27-17-16-22/h20,23H,2-19,22H2,1H3,(H,25,26) |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | (2-aminoethoxy)[2-(hexadecanoyloxy)-3-hydroxypropoxy]phosphinic acid |
|---|
|
Traditional IUPAC Name: |
2-aminoethoxy(2-(hexadecanoyloxy)-3-hydroxypropoxy)phosphinic acid |
|---|
| SMILES: | CCCCCCCCCCCCCCCC(=O)OC(CO)COP(O)(=O)OCCN |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as 2-acyl-sn-glycero-3-phosphoethanolamines. These are glycerophoethanolamines in which the glycerol is esterified with a fatty acid at O-2 position, and linked at position 3 to a phosphoethanolamine. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Glycerophospholipids |
|---|
| Sub Class | Glycerophosphoethanolamines |
|---|
|
Direct Parent |
2-acyl-sn-glycero-3-phosphoethanolamines |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- 2-monoacyl-sn-glycero-3-phosphoethanolamine
- Phosphoethanolamine
- Dialkyl phosphate
- Fatty acid ester
- Fatty acyl
- Alkyl phosphate
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Organic phosphate
- Carboxylic acid ester
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Hydrocarbon derivative
- Primary amine
- Primary alcohol
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Carbonyl group
- Amine
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | 0 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Membrane |
|---|
| Reactions: | |
|---|
|
Pathways: |
Not Available |
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | 46173130 | | Kegg ID | Not Available | | ChemSpider ID | 24608312 | | Wikipedia ID | Not Available | | BioCyc ID | CPD0-2177 | | EcoCyc ID | CPD0-2177 |
|
|---|