|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB000904 |
|---|
|
Identification |
|---|
| Name: |
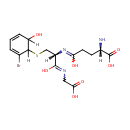
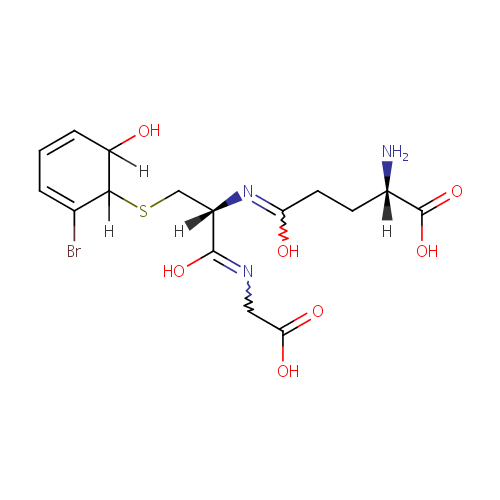
2,3-Dihydro-2-S-glutathionyl-3-hydroxy bromobenzene |
|---|
| Description: | 2,3-dihydro-2-s-glutathionyl-3-hydroxy bromobenzene belongs to the class of Peptides. These are compounds containing an amide derived from two or more amino carboxylic acid molecules (the same or different) by formation of a covalent bond from the carbonyl carbon of one to the nitrogen atom of another. (inferred from compound structure) |
|---|
|
Structure |
|
|---|
| Synonyms: | - (2S)-2-amino-5-(2R)-3-(2-bromo-6-Hydroxycyclohexa-2,4-dien-1-yl)sulfanyl-1-(carboxymethylamino)-1-oxopropan-2-ylamino-5-oxopentanoate
- (2S)-2-amino-5-(2R)-3-(2-bromo-6-hydroxycyclohexa-2,4-dien-1-yl)sulfanyl-1-(carboxymethylamino)-1-oxopropan-2-ylamino-5-oxopentanoic acid
- (2S)-2-amino-5-(2R)-3-(2-bromo-6-Hydroxycyclohexa-2,4-dien-1-yl)sulphanyl-1-(carboxymethylamino)-1-oxopropan-2-ylamino-5-oxopentanoate
- (2S)-2-amino-5-(2R)-3-(2-bromo-6-Hydroxycyclohexa-2,4-dien-1-yl)sulphanyl-1-(carboxymethylamino)-1-oxopropan-2-ylamino-5-oxopentanoic acid
- L-g-Glutamyl-S-(2-bromo-6-hydroxy-2,4-cyclohexadien-1-yl)-L-cysteinylglycine
- L-gamma-Glutamyl-S-(2-bromo-6-hydroxy-2,4-cyclohexadien-1-yl)-L-cysteinylglycine
- L-γ-Glutamyl-S-(2-bromo-6-hydroxy-2,4-cyclohexadien-1-yl)-L-cysteinylglycine
|
|---|
|
Chemical Formula: |
C16H22BrN3O7S |
|---|
| Average Molecular Weight: |
480.331 |
|---|
| Monoisotopic Molecular
Weight: |
479.03618341 |
|---|
| InChI Key: |
BYHFLDCNLZPMDY-PXAOEZFJSA-N |
|---|
| InChI: | InChI=1S/C16H22BrN3O7S/c17-8-2-1-3-11(21)14(8)28-7-10(15(25)19-6-13(23)24)20-12(22)5-4-9(18)16(26)27/h1-3,9-11,14,21H,4-7,18H2,(H,19,25)(H,20,22)(H,23,24)(H,26,27)/t9-,10-,11?,14?/m0/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | (2S)-2-amino-4-{[(1R)-2-[(2-bromo-6-hydroxycyclohexa-2,4-dien-1-yl)sulfanyl]-1-[(carboxymethyl)-C-hydroxycarbonimidoyl]ethyl]-C-hydroxycarbonimidoyl}butanoic acid |
|---|
|
Traditional IUPAC Name: |
C16H22brN3O7S |
|---|
| SMILES: | [H][C@](N)(CCC(O)=N[C@@]([H])(CSC1([H])C(Br)=CC=CC1([H])O)C(O)=NCC(O)=O)C(O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as peptides. These are compounds containing an amide derived from two or more amino carboxylic acid molecules (the same or different) by formation of a covalent bond from the carbonyl carbon of one to the nitrogen atom of another. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
|
Class |
Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
|
Direct Parent |
Peptides |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Alpha peptide
- N-acyl-aliphatic-alpha amino acid
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- D-alpha-amino acid
- Alpha-amino acid or derivatives
- Alpha-amino acid
- Carbocyclic fatty acid
- Halogenated fatty acid
- Amino fatty acid
- Fatty acyl
- Dicarboxylic acid or derivatives
- Cyclic alcohol
- Secondary alcohol
- Bromoalkene
- Haloalkene
- Dialkylthioether
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Sulfenyl compound
- Vinyl halide
- Vinyl bromide
- Thioether
- Carboxylic acid
- Carboximidic acid derivative
- Carboximidic acid
- Hydrocarbon derivative
- Primary amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Organobromide
- Organohalogen compound
- Primary aliphatic amine
- Carbonyl group
- Amine
- Alcohol
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework |
Aliphatic homomonocyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -2 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | HMDB60358 | | Pubchem Compound ID | 11954067 | | Kegg ID | C14848 | | ChemSpider ID | 10128362 | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|