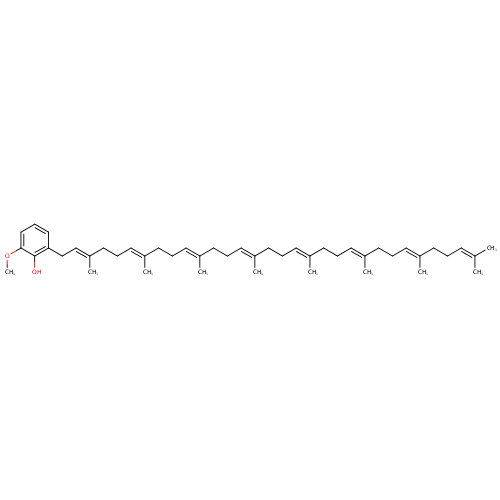2-Octaprenyl-6-methoxyphenol (PAMDB000621)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000621 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | 2-Octaprenyl-6-methoxyphenol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | 2-octaprenyl-6-methoxyphenol belongs to the class of Tetraterpenes. These are terpene molecules containing 10 consecutively linked isoprene units. (inferred from compound structure)2-octaprenyl-6-methoxyphenol is invovled in Ubiquinone and other terpenoid-quinone biosynthesis, and Biosynthesis of secondary metabolites. (KEGG) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C47H72O2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 669.0734 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 668.553231548 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | MARGKPIMNMASKJ-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C47H72O2/c1-37(2)19-11-20-38(3)21-12-22-39(4)23-13-24-40(5)25-14-26-41(6)27-15-28-42(7)29-16-30-43(8)31-17-32-44(9)35-36-45-33-18-34-46(49-10)47(45)48/h18-19,21,23,25,27,29,31,33-35,48H,11-17,20,22,24,26,28,30,32,36H2,1-10H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | 2-methoxy-6-[(2E,6E,10E,14E,18E,22E,26E)-3,7,11,15,19,23,27,31-octamethyldotriaconta-2,6,10,14,18,22,26,30-octaen-1-yl]phenol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | 2-octaprenyl-6-methoxyphenol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | COC1=CC=CC(CC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)C)=C1O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Membrane | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | 2-Octaprenyl-6-hydroxyphenol + S-Adenosylmethionine > 2-Octaprenyl-6-methoxyphenol + S-Adenosylhomocysteine + Hydrogen ion 2-Octaprenyl-6-methoxyphenol + Oxygen > 2-Octaprenyl-6-methoxy-1,4-benzoquinol 2-Octaprenyl-6-hydroxyphenol + S-Adenosylmethionine <> 2-Octaprenyl-6-methoxyphenol + S-Adenosylhomocysteine 2-Octaprenyl-6-methoxyphenol + Oxygen + NADPH + Hydrogen ion > 2-Octaprenyl-6-methoxy-1,4-benzoquinol + Water + NADP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Enzymes
- General function:
- Involved in 2-polyprenyl-6-methoxy-1,4-benzoquinone methyltransferase activity
- Specific function:
- S-adenosyl-L-methionine + 3- demethylubiquinone-9 = S-adenosyl-L-homocysteine + ubiquinone-9
- Gene Name:
- ubiG
- Locus Tag:
- PA3171
- Molecular weight:
- 25.9 kDa
Reactions
| S-adenosyl-L-methionine + 3-demethylubiquinone-n = S-adenosyl-L-homocysteine + ubiquinone-n. |
| S-adenosyl-L-methionine + 3-(all-trans-polyprenyl)benzene-1,2-diol = S-adenosyl-L-homocysteine + 2-methoxy-6-(all-trans-polyprenyl)phenol. |
- General function:
- Involved in oxidoreductase activity
- Specific function:
- Oxygenase that introduces the hydroxyl group at carbon four of 2-octaprenyl-6-methoxyphenol resulting in the formation of 2-octaprenyl-6-methoxy-1,4-benzoquinone
- Gene Name:
- ubiH
- Locus Tag:
- PA5223
- Molecular weight:
- 43 kDa