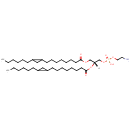
Search Results for compounds
Searching compounds for
returned 4373 results.
Displaying compounds 1071 - 1080 of
4373 in total
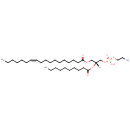
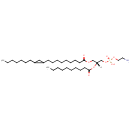
PE(18:1(11Z)/10:0) (PAMDB002021)
IUPAC:
(2-aminoethoxy)[(2R)-2-(decanoyloxy)-3-[(11Z)-octadec-11-enoyloxy]propoxy]phosphinic acid
CAS: Not Available
Description: PE(18:1(11Z)/10:0) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. PE(18:1(11Z)/10:0), in particular, consists of one 11Z-octadecenoyl chain to the C-1 atom, and one decanoyl to the C-2 atom. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
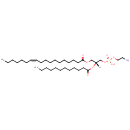
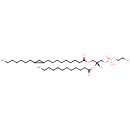
PE(18:1(11Z)/12:0) (PAMDB002022)
IUPAC:
(2-aminoethoxy)[(2R)-2-(dodecanoyloxy)-3-[(11Z)-octadec-11-enoyloxy]propoxy]phosphinic acid
CAS: Not Available
Description: PE(18:1(11Z)/12:0) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. PE(18:1(11Z)/12:0), in particular, consists of one 11Z-octadecenoyl chain to the C-1 atom, and one dodecanoyl to the C-2 atom. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
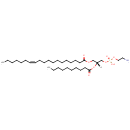
PE(19:1(12Z)/10:0) (PAMDB002023)
IUPAC:
(2-aminoethoxy)[(2R)-2-(decanoyloxy)-3-[(12Z)-nonadec-12-enoyloxy]propoxy]phosphinic acid
CAS: Not Available
Description: PE(19:1(12Z)/10:0) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. PE(19:1(12Z)/10:0), in particular, consists of one 12Z-nonadecenoyl chain to the C-1 atom, and one decanoyl to the C-2 atom. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
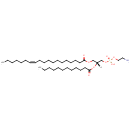
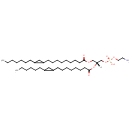
PE(19:1(12Z)/12:0) (PAMDB002024)
IUPAC:
(2-aminoethoxy)[(2R)-2-(dodecanoyloxy)-3-[(12Z)-nonadec-12-enoyloxy]propoxy]phosphinic acid
CAS: Not Available
Description: PE(19:1(12Z)/12:0) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. PE(19:1(12Z)/12:0), in particular, consists of one 12Z-nonadecenoyl chain to the C-1 atom, and one dodecanoyl to the C-2 atom. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
PE(19:0cycw8c/10:0) (PAMDB002025)
IUPAC:
(2-aminoethoxy)[(2R)-2-(decanoyloxy)-3-{[9-(2-heptylcyclopropyl)nonanoyl]oxy}propoxy]phosphinic acid
CAS: Not Available
Description: PE(19:0cycw8c/10:0) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. PE(19:0cycw8c/10:0), in particular, consists of one 9-(2-heptylcyclopropyl)nonanoyl chain to the C-1 atom, and one decanoyl to the C-2 atom. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
PE(19:0cycw8c/12:0) (PAMDB002026)
IUPAC:
(2-aminoethoxy)[(2R)-2-(dodecanoyloxy)-3-{[9-(2-heptylcyclopropyl)nonanoyl]oxy}propoxy]phosphinic acid
CAS: Not Available
Description: PE(19:0cycw8c/12:0) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. PE(19:0cycw8c/12:0), in particular, consists of one 9-(2-heptylcyclopropyl)nonanoyl chain to the C-1 atom, and one dodecanoyl to the C-2 atom. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
PE(17:0cycw7c/17:0cycw7c) (PAMDB002027)
IUPAC:
(2-aminoethoxy)[(2R)-2,3-bis({[8-(2-hexylcyclopropyl)octanoyl]oxy})propoxy]phosphinic acid
CAS: Not Available
Description: PE(17:0cycw7c/17:0cycw7c) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 positions. PE(17:0cycw7c/17:0cycw7c), in particular, consists of two heptadec-9-10-cyclo-anoyl chains at positions C-1 and C-2. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
PE(19:0cycw8c/17:0cycw7c) (PAMDB002028)
IUPAC:
(2-aminoethoxy)[(2R)-3-{[9-(2-heptylcyclopropyl)nonanoyl]oxy}-2-{[8-(2-hexylcyclopropyl)octanoyl]oxy}propoxy]phosphinic acid
CAS: Not Available
Description: PE(19:0cycw8c/17:0cycw7c) is a phosphatidylethanolamine. It is a glycerophospholipid in which a phosphorylethanolamine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, glycerophosphoethanolamines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 atoms. PE(19:0cycw8c/17:0cycw7c), in particular, consists of one 9-(2-heptylcyclopropyl)nonanoyl chain to the C-1 atom, and one heptadec-9-10-cyclo-anoyl to the C-2 atom. While most phospholipids have a saturated fatty acid on C-1 and an unsaturated fatty acid on C-2 of the glycerol backbone, the fatty acid distribution at the C-1 and C-2 positions of glycerol within phospholipids is continually in flux, owing to phospholipid degradation and the continuous phospholipid remodeling that occurs while these molecules are in membranes. PEs are neutral zwitterions at physiological pH. They mostly have palmitic or stearic acid on carbon 1 and a long chain unsaturated fatty acid (e.g. 18:2, 20:4 and 22:6) on carbon 2. PE synthesis can occur via two pathways. The first requires that ethanolamine be activated by phosphorylation and then coupled to CDP. The ethanolamine is then transferred from CDP-ethanolamine to phosphatidic acid to yield PE. The second involves the decarboxylation of PS.
PG(14:0/17:0cycw7c) (PAMDB002029)
IUPAC:
[(2S)-2,3-dihydroxypropoxy][(2R)-3-{[8-(2-hexylcyclopropyl)octanoyl]oxy}-2-(tetradecanoyloxy)propoxy]phosphinic acid
CAS: Not Available
Description: PG(14:0/17:0cycw7c) is a phosphatidylglycerol. Phosphatidylglycerols consist of a glycerol 3-phosphate backbone esterified to either saturated or unsaturated fatty acids on carbons 1 and 2. As is the case with diacylglycerols, phosphatidylglycerols can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 positions. PG(14:0/17:0cycw7c), in particular, consists of one tetradecanoyl chain to the C-1 atom, and one heptadec-9-10-cyclo-anoyl to the C-2 atom. In Pseudomonas aeruginosa glycerophospholipid metabolism, phosphatidylglycerol is formed from phosphatidic acid (1,2-diacyl-sn-glycerol 3-phosphate) by a sequence of enzymatic reactions that proceeds via two intermediates, cytidine diphosphate diacylglycerol (CDP-diacylglycerol) and phosphatidylglycerophosphate (PGP, a phosphorylated phosphatidylglycerol). Phosphatidylglycerols, along with CDP-diacylglycerol, also serve as precursor molecules for the synthesis of cardiolipin, a phospholipid found in membranes.
PG(15:0/16:1) (PAMDB002030)
IUPAC:
(2,3-dihydroxypropoxy)({2-[(9Z)-hexadec-9-enoyloxy]-3-(pentadecanoyloxy)propoxy})phosphinic acid
CAS: Not Available
Description: PG(15:0/16:1) is a phosphatidylglycerol or glycerophospholipid (PG or GP). It is a glycerophospholipid in which a phosphoglycerol moiety occupies a glycerol substitution site. As is the case with diacylglycerols, phosphatidylglycerols can have many different combinations of fatty acids of varying lengths and saturation attached at the C-1 and C-2 positions. Fatty acids containing 16, 18 and 20 carbons are the most common. PG(15:0/16:1), in particular, consists of one chain of pentadecanoic acid at the C-1 position and one chain of palmitoleic acid at the C-2 position. Phosphatidylglycerol is formed from phosphatidic acid by a sequence of enzymatic reactions that proceeds via the intermediate, cytidine diphosphate diacylglycerol (CDP-diacylglycerol). Bioynthesis proceeds by condensation of phosphatidic acid and cytidine triphosphate with elimination of pyrophosphate via the action of phosphatidate cytidyltransferase (or CDP-synthase). CDP-diacylglycerol then reacts with glycerol-3-phosphate via phosphatidylglycerophosphate synthase to form 3-sn-phosphatidyl-1'-sn-glycerol 3'-phosphoric acid, with the release of cytidine monophosphate (CMP). Finally, phosphatidylglycerol is formed by the action of specific phosphatases. PG also serves as a precursor for the synthesis of cardiolipin. PG is synthesized from CDP-diacylglycerol and glycerol-3-phosphate.