|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB120513 |
|---|
|
Identification |
|---|
| Name: |
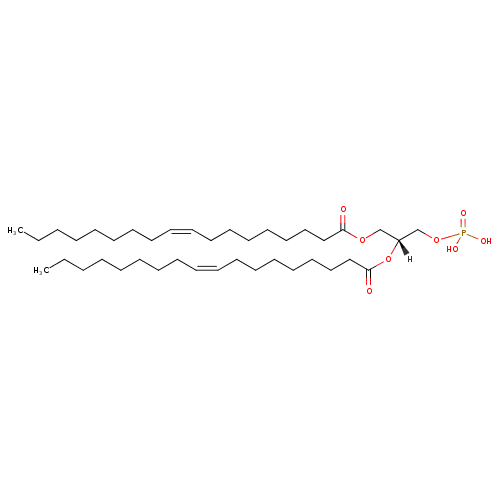
1-18:1-2-18:1-phosphatidate |
|---|
| Description: | PA(18:1(9Z)/18:1(9Z)) is a phosphatidic acid. It is a glycerophospholipid in which a phosphate moiety occupies a glycerol substitution site. As is the case with diacylglycerols, phosphatidic acids can have many different combinations of fatty acids of varying lengths and saturation attached at the C-1 and C-2 positions. Fatty acids containing 16, 18 and 20 carbons are the most common. PA(18:1(9Z)/18:1(9Z)), in particular, consists of one chain of oleic acid at the C-1 position and one chain of oleic acid at the C-2 position. The oleic acid moiety is derived from vegetable oils, especially olive and canola oil, while the oleic acid moiety is derived from vegetable oils, especially olive and canola oil. Phosphatidic acids are quite rare but are extremely important as intermediates in the biosynthesis of triacylglycerols and phospholipids. Indeed, the concentration of phosphatidic acids is often over-estimated in tissues and biofluids as it can arise by inadvertent enzymatic hydrolysis during inappropriate storage or extraction conditions during analysis. The main biosynthetic route of phosphatidic acid in animal tissues involves sequential acylation of alpha-glycerophosphate by acyl-CoA derivatives of fatty acids. PAs are biologically active lipids that can stimulate a large range of responses in many different cell types, such as platelet aggregation, smooth muscle contraction, in vivo vasoactive effects, chemotaxis, expression of adhesion molecules, increased tight junction permeability of endothelial cells, induction of stress fibres, modulation of cardiac contractility, and many others. Diacylglycerols (DAGs) can be converted to PAs by DAG kinases and indirect evidence supports the notion that PAs alter the excitability of neurons. Phospholipase Ds (PLDs), which catalyze the conversion of glycerolphospholipids, particularly phosphatidylcholine, to PAs and the conversion of N-arachidonoyl-phosphatidylethanolamine (NAPE) to anandamide and PAs are activated by several inflammatory mediators including bradykinin, ATP and glutamate. PAs activate downstream signaling pathways such as PKCs and mitogen-activated protein kinases (MAPKs), which are linked to an increase in sensitivity of sensory neurons either during inflammation or in chronic pain models. Circumstantial evidence that PAs are converted to DAGs. (PMID: 12618218 , 16185776 ). |
|---|
|
Structure |
|
|---|
| Synonyms: | - 18:1-18:1-PA
- 1-18:1-2-18:1-phosphatidic acid
- 1,2-(9Z-octadecenoyl)-sn-glycero-3-phosphate
|
|---|
|
Chemical Formula: |
C39H71O8P |
|---|
| Average Molecular Weight: |
698.959 |
|---|
| Monoisotopic Molecular
Weight: |
700.50433 |
|---|
| InChI Key: |
MHUWZNTUIIFHAS-CLFAGFIQSA-L |
|---|
| InChI: | InChI=1S/C39H73O8P/c1-3-5-7-9-11-13-15-17-19-21-23-25-27-29-31-33-38(40)45-35-37(36-46-48(42,43)44)47-39(41)34-32-30-28-26-24-22-20-18-16-14-12-10-8-6-4-2/h17-20,37H,3-16,21-36H2,1-2H3,(H2,42,43,44)/p-2/b19-17-,20-18- |
|---|
| CAS
number: |
14268-17-8 |
|---|
| IUPAC Name: | [(2R)-2,3-bis[(9Z)-octadec-9-enoyloxy]propoxy]phosphonic acid |
|---|
|
Traditional IUPAC Name: |
(2R)-2,3-bis[(9Z)-octadec-9-enoyloxy]propoxyphosphonic acid |
|---|
| SMILES: | CCCCCCCCC=CCCCCCCCC(OCC(OC(=O)CCCCCCCC=CCCCCCCCC)COP([O-])([O-])=O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as 1,2-diacylglycerol-3-phosphates. These are glycerol-3-phosphates in which the glycerol moiety is bonded to two aliphatic chains through ester linkages. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Lipids and lipid-like molecules |
|---|
| Sub Class | Glycerophospholipids |
|---|
|
Direct Parent |
1,2-diacylglycerol-3-phosphates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- 1,2-diacylglycerol-3-phosphate
- Fatty acid ester
- Monoalkyl phosphate
- Dicarboxylic acid or derivatives
- Fatty acyl
- Alkyl phosphate
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Carboxylic acid ester
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Organooxygen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | Not Available |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
- superpathway phosphatidate biosynthesis (yeast)PWY-7411
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Niedernberg A, Tunaru S, Blaukat A, Ardati A, Kostenis E: Sphingosine 1-phosphate and dioleoylphosphatidic acid are low affinity agonists for the orphan receptor GPR63. Cell Signal. 2003 Apr;15(4):435-46. [12618218 ]
- Park KA, Vasko MR: Lipid mediators of sensitivity in sensory neurons. Trends Pharmacol Sci. 2005 Nov;26(11):571-7. Epub 2005 Sep 26. [16185776 ]
|
|---|
| Synthesis Reference: |
Hermetter, A.; Paltauf, F.; Hauser, H. Synthesis of diacyl and alkylacyl glycerophosphoserines. Chemistry and Physics of Lipids (1982), 30(1), 35-45. |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|