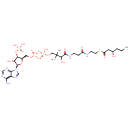|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB120480 |
|---|
|
Identification |
|---|
| Name: |
(S)-3-hydroxyhexanoyl-CoA |
|---|
| Description: | A hydroxy fatty acyl-CoA that results from the formal condensation of the thiol group of coenzyme A with the carboxy group of (S)-3-hydroxyhexanoyl-CoA. |
|---|
|
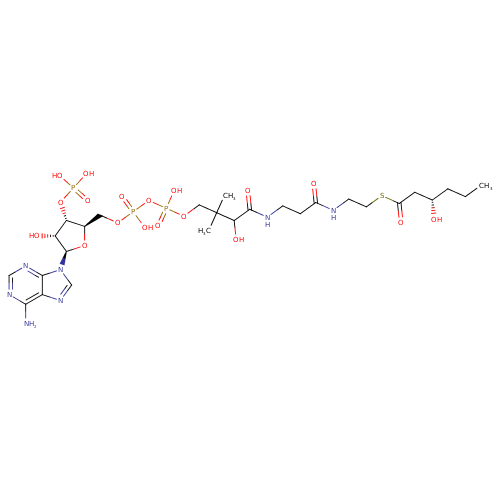
Structure |
|
|---|
| Synonyms: | - (S)-3-hydroxycaproyl-CoA
- (S)-3-hydroxycaproyl-coenzyme A
- (S)-3-Hydroxyhexanoyl-CoA
- (S)-Hydroxyhexanoyl-CoA
- 3-hydroxyhexanoyl-coenzyme A
|
|---|
|
Chemical Formula: |
C27H42N7O18P3S |
|---|
| Average Molecular Weight: |
877.646 |
|---|
| Monoisotopic Molecular
Weight: |
881.1833 |
|---|
| InChI Key: |
VAAHKRMGOFIORX-DWUFXMDISA-J |
|---|
| InChI: | InChI=1S/C27H46N7O18P3S/c1-4-5-15(35)10-18(37)56-9-8-29-17(36)6-7-30-25(40)22(39)27(2,3)12-49-55(46,47)52-54(44,45)48-11-16-21(51-53(41,42)43)20(38)26(50-16)34-14-33-19-23(28)31-13-32-24(19)34/h13-16,20-22,26,35,38-39H,4-12H2,1-3H3,(H,29,36)(H,30,40)(H,44,45)(H,46,47)(H2,28,31,32)(H2,41,42,43)/p-4/t15-,16+,20+,21+,22?,26+/m0/s1 |
|---|
| CAS
number: |
5060-32-2 |
|---|
| IUPAC Name: | 3'- phosphoadenosine 5'- phosphoadenosine 5'- {3- {3- [(3R)- [(3R)- 3- 3- hydroxy- hydroxy- 4- 4- ({3- ({3- [(2- [(2- {[(3S)- {[(3S)- 3- 3- hydroxyhexanoyl]sulfanyl}ethyl)amino]- hydroxyhexanoyl]sulfanyl}ethyl)amino]- 3- 3- oxopropyl}amino)- oxopropyl}amino)- 2,2- 2,2- dimethyl- dimethyl- 4- 4- oxobutyl] dihydrogen diphosphate} oxobutyl] dihydrogen diphosphate} |
|---|
|
Traditional IUPAC Name: |
[(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-4-hydroxy-2-[({hydroxy[hydroxy(3-hydroxy-3-({2-[(2-{[(3S)-3-hydroxyhexanoyl]sulfanyl}ethyl)carbamoyl]ethyl}carbamoyl)-2,2-dimethylpropoxy)phosphoryl]oxyphosphoryl}oxy)methyl]oxolan-3-yl]oxyphosphonic acid |
|---|
| SMILES: | CCCC(CC(SCCNC(CCNC(C(C(COP(=O)([O-])OP(OCC1(OC(C(C1OP([O-])([O-])=O)O)N3(C=NC2(C(=NC=NC=23)N))))([O-])=O)(C)C)O)=O)=O)=O)O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as (s)-3-hydroxyacyl coas. These are organic compounds containing a (S)-3-hydroxyl acylated coenzyme A derivative. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Lipids and lipid-like molecules |
|---|
| Sub Class | Fatty Acyls |
|---|
|
Direct Parent |
(S)-3-hydroxyacyl CoAs |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Coenzyme a or derivatives
- Purine ribonucleoside 3',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Pentose phosphate
- Pentose-5-phosphate
- Ribonucleoside 3'-phosphate
- Beta amino acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Organic pyrophosphate
- 6-aminopurine
- Pentose monosaccharide
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Aminopyrimidine
- Fatty amide
- Imidolactam
- Monosaccharide
- N-acyl-amine
- N-substituted imidazole
- Organic phosphoric acid derivative
- Alkyl phosphate
- Phosphoric acid ester
- Primary aromatic amine
- Pyrimidine
- Oxolane
- Azole
- Imidazole
- Heteroaromatic compound
- Amino acid or derivatives
- Thiocarboxylic acid ester
- Carboxamide group
- Carbothioic s-ester
- Secondary carboxylic acid amide
- Secondary alcohol
- Sulfenyl compound
- Thiocarboxylic acid or derivatives
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Carboxylic acid derivative
- Hydrocarbon derivative
- Alcohol
- Organic nitrogen compound
- Amine
- Organonitrogen compound
- Carbonyl group
- Organooxygen compound
- Organosulfur compound
- Organic oxygen compound
- Organopnictogen compound
- Primary amine
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework |
Aromatic heteropolycyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | 0 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | -3.019 | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Dalluge JJ, Gort S, Hobson R, Selifonova O, Amore F, Gokarn R: Separation and identification of organic acid-coenzyme A thioesters using liquid chromatography/electrospray ionization-mass spectrometry. Anal Bioanal Chem. 2002 Nov;374(5):835-40. Epub 2002 Oct 8. [12434239 ]
- Klees AG, Linder D, Buckel W: 2-Hydroxyglutaryl-CoA dehydratase from Fusobacterium nucleatum (subsp. nucleatum): an iron-sulfur flavoprotein. Arch Microbiol. 1992;158(4):294-301. [1417419 ]
- Kim J, Hetzel M, Boiangiu CD, Buckel W: Dehydration of (R)-2-hydroxyacyl-CoA to enoyl-CoA in the fermentation of alpha-amino acids by anaerobic bacteria. FEMS Microbiol Rev. 2004 Oct;28(4):455-68. [15374661 ]
- Hofmeister AE, Buckel W: (R)-lactyl-CoA dehydratase from Clostridium propionicum. Stereochemistry of the dehydration of (R)-2-hydroxybutyryl-CoA to crotonyl-CoA. Eur J Biochem. 1992 Jun 1;206(2):547-52. [1597194 ]
- Kuchta RD, Hanson GR, Holmquist B, Abeles RH: Fe-S centers in lactyl-CoA dehydratase. Biochemistry. 1986 Nov 18;25(23):7301-7. [3026450 ]
- Kuchta RD, Abeles RH: Lactate reduction in Clostridium propionicum. Purification and properties of lactyl-CoA dehydratase. J Biol Chem. 1985 Oct 25;260(24):13181-9. [4055736 ]
- Sokatch JR: Alanine and aspartate formation during growth on valine-C14 by Pseudomonas aeruginosa. J Bacteriol. 1966 Jul;92(1):72-5. [4957438 ]
- Megraw RE, Reeves HC, Ajl SJ: Formation of lactyl-coenzyme A and pyruvyl-coenzyme A from lactic acid by Escherichia coli. J Bacteriol. 1965 Oct;90(4):984-8. [5321404 ]
|
|---|
| Synthesis Reference: |
Tamvakopoulos, Constantine S.; Anderson, Vernon E. Detection of acyl-coenzyme A thioester intermediates of fatty acid b-oxidation as the N-acylglycines by negative-ion chemical ionization gas chromatography mass spectrometry. Analytical Biochemistry |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|