|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB120380 |
|---|
|
Identification |
|---|
| Name: |
linamarin |
|---|
| Description: | Linamarin is found in coffee and coffee products. Linamarin occurs in manioc (Manihot utilissimus), flax (Linum usitatissimum), Phaseolus lunatus (butter bean), Trifolium repens (white clover) and other plants. First isloated in 1830. |
|---|
|
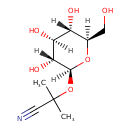
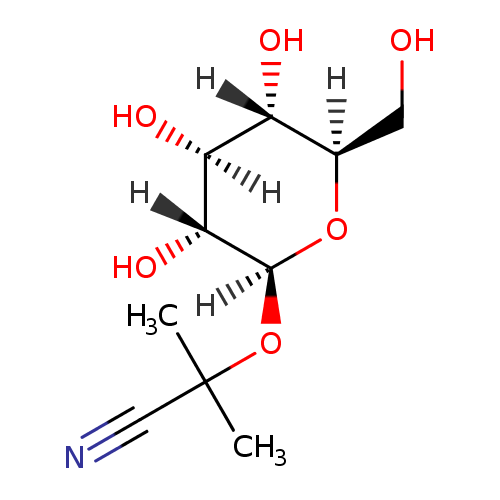
Structure |
|
|---|
| Synonyms: | - 1-cyano-1-methylethyl beta-D-glucoside
- Linamarin
- linamarin
- Phaseolunatin
|
|---|
|
Chemical Formula: |
C10H17NO6 |
|---|
| Average Molecular Weight: |
247.247 |
|---|
| Monoisotopic Molecular
Weight: |
247.10559 |
|---|
| InChI Key: |
QLTCHMYAEJEXBT-ZEBDFXRSSA-N |
|---|
| InChI: | InChI=1S/C10H17NO6/c1-10(2,4-11)17-9-8(15)7(14)6(13)5(3-12)16-9/h5-9,12-15H,3H2,1-2H3/t5-,6-,7+,8-,9+/m1/s1 |
|---|
| CAS
number: |
554-35-8 |
|---|
| IUPAC Name: | 2-(β-D-glucopyranosyloxy)-2-methylpropanenitrile |
|---|
|
Traditional IUPAC Name: |
linamarin |
|---|
| SMILES: | CC(C)(C#N)OC1(OC(CO)C(O)C(O)C(O)1) |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as cyanogenic glycosides. These are glycosides in which the aglycone moiety contains a cyanide group. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Organic oxygen compounds |
|---|
| Sub Class | Organooxygen compounds |
|---|
|
Direct Parent |
Cyanogenic glycosides |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Cyanogenic glycoside
- Hexose monosaccharide
- O-glycosyl compound
- Monosaccharide
- Oxane
- Secondary alcohol
- Polyol
- Organoheterocyclic compound
- Oxacycle
- Nitrile
- Carbonitrile
- Acetal
- Primary alcohol
- Organonitrogen compound
- Hydrocarbon derivative
- Organopnictogen compound
- Organic nitrogen compound
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors |
- beta-D-glucoside (CHEBI:16441 )
- Cyanogenic glucosides derived from valine or isoleucine (C01594 )
|
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | 0 |
|---|
|
Melting point: |
143 - 144 °C |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | 143 - 144 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Yannai, Shmuel. (2004) Dictionary of food compounds with CD-ROM: Additives, flavors, and ingredients. Boca Raton: Chapman & Hall/CRC.
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|