|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB120332 |
|---|
|
Identification |
|---|
| Name: |
hexanoyl-CoA |
|---|
| Description: | A medium-chain fatty acyl-CoA having hexanoyl as the S-acyl group. |
|---|
|
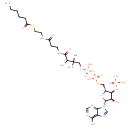
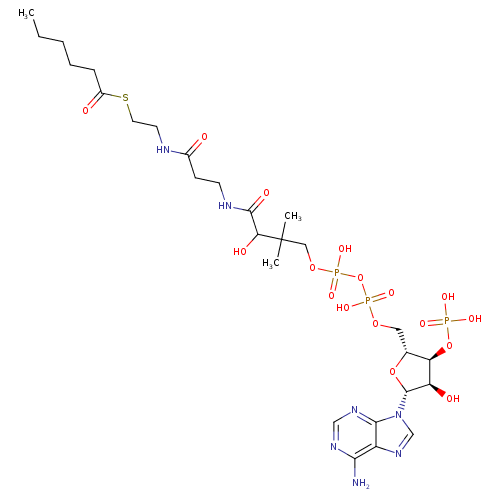
Structure |
|
|---|
| Synonyms: | - caproyl-CoA
- caproyl-coenzyme A
- Coenzyme A, S-hexanoate
- Hexanoyl-CoA
- hexanoyl-CoA
- Hexanoyl-coenzyme A
- n-hexanoyl-CoA
- n-hexanoyl-coenzyme A
- S-hexanoyl-CoA
- S-Hexanoyl-coenzym-A
- S-hexanoyl-coenzyme-A
|
|---|
|
Chemical Formula: |
C27H42N7O17P3S |
|---|
| Average Molecular Weight: |
861.647 |
|---|
| Monoisotopic Molecular
Weight: |
865.18835 |
|---|
| InChI Key: |
OEXFMSFODMQEPE-UHFFFAOYSA-J |
|---|
| InChI: | InChI=1S/C27H46N7O17P3S/c1-4-5-6-7-18(36)55-11-10-29-17(35)8-9-30-25(39)22(38)27(2,3)13-48-54(45,46)51-53(43,44)47-12-16-21(50-52(40,41)42)20(37)26(49-16)34-15-33-19-23(28)31-14-32-24(19)34/h14-16,20-22,26,37-38H,4-13H2,1-3H3,(H,29,35)(H,30,39)(H,43,44)(H,45,46)(H2,28,31,32)(H2,40,41,42)/p-4 |
|---|
| CAS
number: |
5060-32-2 |
|---|
| IUPAC Name: | 3'- phosphoadenosine 5'- phosphoadenosine 5'- (3- (3- {(3R)- {(3R)- 3- 3- hydroxy- hydroxy- 4- 4- [(3- [(3- {[2- {[2- (hexanoylsulfanyl)ethyl]amino}- (hexanoylsulfanyl)ethyl]amino}- 3- 3- oxopropyl)amino]- oxopropyl)amino]- 2,2- 2,2- dimethyl- dimethyl- 4- 4- oxobutyl} dihydrogen diphosphate) oxobutyl} dihydrogen diphosphate) |
|---|
|
Traditional IUPAC Name: |
[(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-2-{[({3-[(2-{[2-(hexanoylsulfanyl)ethyl]carbamoyl}ethyl)carbamoyl]-3-hydroxy-2,2-dimethylpropoxy(hydroxy)phosphoryl}oxy(hydroxy)phosphoryl)oxy]methyl}-4-hydroxyoxolan-3-yl]oxyphosphonic acid |
|---|
| SMILES: | CCCCCC(=O)SCCNC(CCNC(C(C(COP(OP(OCC3(C(OP(=O)([O-])[O-])C(O)C(N2(C1(N=CN=C(N)C=1N=C2)))O3))(=O)[O-])(=O)[O-])(C)C)O)=O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as 2,3,4-saturated fatty acyl coas. These are acyl-CoAs carrying a 2,3,4-saturated fatty acyl chain. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Lipids and lipid-like molecules |
|---|
| Sub Class | Fatty Acyls |
|---|
|
Direct Parent |
2,3,4-saturated fatty acyl CoAs |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Coenzyme a or derivatives
- Purine ribonucleoside 3',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Pentose phosphate
- Pentose-5-phosphate
- Ribonucleoside 3'-phosphate
- Beta amino acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Organic pyrophosphate
- 6-aminopurine
- Pentose monosaccharide
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Aminopyrimidine
- Fatty amide
- Imidolactam
- Monosaccharide
- N-acyl-amine
- N-substituted imidazole
- Organic phosphoric acid derivative
- Alkyl phosphate
- Phosphoric acid ester
- Primary aromatic amine
- Pyrimidine
- Oxolane
- Azole
- Imidazole
- Heteroaromatic compound
- Amino acid or derivatives
- Thiocarboxylic acid ester
- Carboxamide group
- Carbothioic s-ester
- Secondary carboxylic acid amide
- Secondary alcohol
- Sulfenyl compound
- Thiocarboxylic acid or derivatives
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Carboxylic acid derivative
- Hydrocarbon derivative
- Alcohol
- Organic nitrogen compound
- Amine
- Organonitrogen compound
- Carbonyl group
- Organooxygen compound
- Organosulfur compound
- Organic oxygen compound
- Organopnictogen compound
- Primary amine
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework |
Aromatic heteropolycyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | 0 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Ohgusu H, Shirouzu K, Nakamura Y, Nakashima Y, Ida T, Sato T, Kojima M (2009)Ghrelin O-acyltransferase (GOAT) has a preference for n-hexanoyl-CoA over n-octanoyl-CoA as an acyl donor. Biochemical and biophysical research communications 386, Pubmed: 19501572
- Crawford JM, Vagstad AL, Ehrlich KC, Townsend CA (2008)Starter unit specificity directs genome mining of polyketide synthase pathways in fungi. Bioorganic chemistry 36, Pubmed: 18215412
- Kasuya F, Kazumi M, Tatsuki T, Suzuki R (2009)Effect of salicylic acid and diclofenac on the medium-chain and long-chain acyl-CoA formation in the liver and brain of mouse. Journal of applied toxicology : JAT 29, Pubmed: 19391105
- Niezen-Koning KE, Wanders RJ, Nagel GT, Sewell AC, Heymans HS (1994)Measurement of short-chain acyl-CoA dehydrogenase (SCAD) in cultured skin fibroblasts with hexanoyl-CoA as a competitive inhibitor to eliminate the contribution of medium-chain acyl-CoA dehydrogenase. Clinica chimica acta; international journal of clinical chemistry 229, Pubmed: 7988059
- Hansen JK, Knudsen J (1980)Transacylation as a chain-termination mechanism in fatty acid synthesis by mammalian fatty acid synthetase. Synthesis of butyrate and hexanoate by lactating cow mammary gland fatty acid synthetase. The Biochemical journal 186, Pubmed: 7370014
- Marks MD, Tian L, Wenger JP, Omburo SN, Soto-Fuentes W, He J, Gang DR, Weiblen GD, Dixon RA (2009)Identification of candidate genes affecting Delta9-tetrahydrocannabinol biosynthesis in Cannabis sativa. Journal of experimental botany 60, Pubmed: 19581347
|
|---|
| Synthesis Reference: |
Pullman, Maynard E. Convenient and versatile method for the purification of CoA thiol esters. Analytical Biochemistry (1973), 54(1), 188-98. |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|