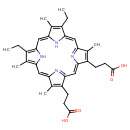
131-hydroxy-magnesium-protoporphyrin IX 13-monomethyl ester (PAMDB120283)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB120283 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | 131-hydroxy-magnesium-protoporphyrin IX 13-monomethyl ester | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | The conjugate base of magnesium 131-hydroxyprotoporphyrin 13-monomethyl ester, formed by deprotonation of the carboxyethyl group at C-17. It is the principal species at pH 7.3. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C35H33N4O5MG | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 613.974 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 614.238 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | MVCDIAGKFJFPBB-JXBSUKTBSA-L | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C35H35N4O5.Mg/c1-8-21-17(3)24-12-25-19(5)23(10-11-33(41)42)30(38-25)15-31-35(32(40)16-34(43)44-7)20(6)27(39-31)14-29-22(9-2)18(4)26(37-29)13-28(21)36-24;/h8-9,12-15,32,40H,1-2,10-11,16H2,3-7H3,(H2-,36,37,38,39,41,42);/q-1;+2/p-2/b24-12-,25-12-,26-13-,27-14-,28-13-,29-14-,30-15-,31-15-; | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 493-90-3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | 3-[20-(2-carboxyethyl)-10,15-diethyl-5,9,14,19-tetramethyl-21,22,23,24-tetraazapentacyclo[16.2.1.1?,??1????.1??,???tetracosa-1,3(24),4,6,8,10,12,14,16,18(21),19-undecaen-4-yl]propanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | MPIX | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | C=CC1(C5(N6(C(C=1C)=CC2(C(=C(C(N=2)=CC3(N(C(=C(C=3C(CC(OC)=O)O)C)C=C4(C(=C(C(=N4)C=5)C)C=C))[Mg]6))CCC(=O)[O-])C)))) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as metalloporphyrins. These are polycyclic compounds containing a porphyrin moiety and a metal atom. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organoheterocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Tetrapyrroles and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Metallotetrapyrroles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Metalloporphyrins | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aromatic heteropolycyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | magnesium-protoporphyrin IX 13-monomethyl ester + Water + NADP+ → 131-hydroxy-magnesium-protoporphyrin IX 13-monomethyl ester + NADPH + Hydrogen ion 131-hydroxy-magnesium-protoporphyrin IX 13-monomethyl ester + NADP+ → 131-oxo-magnesium-protoporphyrin IX 13-monomethyl ester + NADPH + Hydrogen ion magnesium-protoporphyrin IX 13-monomethyl ester + Oxygen + NADPH + Hydrogen ion → 131-hydroxy-magnesium-protoporphyrin IX 13-monomethyl ester + NADP+ + Water 131-hydroxy-magnesium-protoporphyrin IX 13-monomethyl ester + NADPH + Hydrogen ion + Oxygen → 131-oxo-magnesium-protoporphyrin IX 13-monomethyl ester + NADP+ + Water | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Fabry, T. L.; Simo, C.; Javaherian, K. Preparation of mesoporphyrin IX and copper and manganese mesoporphyrin complexes of apohemoglobin. Biochimica et Biophysica Acta, Protein Structure (1968), 160(1), 118-22. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||