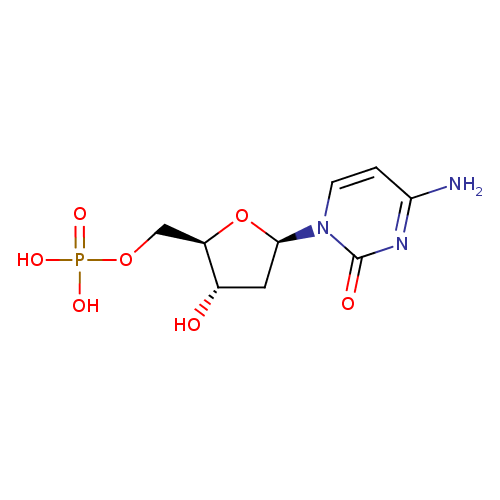
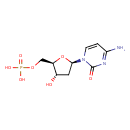| References: |
- Itoh K, Aida S, Ishiwata S, Yamaguchi T, Ishida N, Mizugaki M: Immunochemical detection of urinary 5-methyl-2'-deoxycytidine as a potential biologic marker for leukemia. Clin Chim Acta. 1995 Jan 31;234(1-2):37-45. [7758221 ]
- Mills GC, Schmalstieg FC, Goldblum RM: Urinary excretion of modified purines and nucleosides in immunodeficient children. Biochem Med. 1985 Aug;34(1):37-51. [4052062 ]
- Chen HJ, Wu CF, Hong CL, Chang CM: Urinary excretion of 3,N4-etheno-2'-deoxycytidine in humans as a biomarker of oxidative stress: association with cigarette smoking. Chem Res Toxicol. 2004 Jul;17(7):896-903. [15257614 ]
- Zambonin CG, Aresta A, Palmisano F, Specchia G, Liso V: Liquid chromatographic determination of urinary 5-methyl-2'-deoxycytidine and pseudouridine as potential biological markers for leukaemia. J Pharm Biomed Anal. 1999 Dec;21(5):1045-51. [10703973 ]
- Abbruzzese JL, Grunewald R, Weeks EA, Gravel D, Adams T, Nowak B, Mineishi S, Tarassoff P, Satterlee W, Raber MN, et al.: A phase I clinical, plasma, and cellular pharmacology study of gemcitabine. J Clin Oncol. 1991 Mar;9(3):491-8. [1999720 ]
- Zambonin CG, Palmisano F: Electrospray ionization mass spectrometry of 5-methyl-2'-deoxycytidine and its determination in urine by liquid chromatography/electrospray ionization tandem mass spectrometry. Rapid Commun Mass Spectrom. 1999;13(21):2160-5. [10523775 ]
- Masuda N, Matsui K, Yamamoto N, Nogami T, Nakagawa K, Negoro S, Takeda K, Takifuji N, Yamada M, Kudoh S, Okuda T, Nemoto S, Ogawa K, Myobudani H, Nihira S, Fukuoka M: Phase I trial of oral 2'-deoxy-2'-methylidenecytidine: on a daily x 14-day schedule. Clin Cancer Res. 2000 Jun;6(6):2288-94. [10873079 ]
- Broder S: Pharmacodynamics of 2',3'-dideoxycytidine: an inhibitor of human immunodeficiency virus. Am J Med. 1990 May 21;88(5B):2S-7S. [1692446 ]
- Chen HJ, Lin TC, Hong CL, Chiang LC: Analysis of 3,N(4)-ethenocytosine in DNA and in human urine by isotope dilution gas chromatography/negative ion chemical ionization/mass spectrometry. Chem Res Toxicol. 2001 Dec;14(12):1612-9. [11743744 ]
- Sottani C, Zucchetti M, Zaffaroni M, Bettinelli M, Minoia C: Validated procedure for simultaneous trace level determination of the anti-cancer agent gemcitabine and its metabolite in human urine by high-performance liquid chromatography with tandem mass spectrometry. Rapid Commun Mass Spectrom. 2004;18(10):1017-23. [15150823 ]
- Mazurets AF, Kubyshkin VF, Dziuba MV: [The cytochemical characteristics of the blood polymorphonuclear leukocytes in dilated cardiomyopathy and an inflammatory myocardial lesion] Lik Sprava. 1994 Jul-Aug;(7-8):109-12. [7900329 ]
- Zegers BJ, Stoop JW, Staal GE, Wadman SK: An approach to the restoration of T cell function in a purine nucleoside phosphorylase deficient patient. Ciba Found Symp. 1978;(68):231-53. [115663 ]
- de Bono JS, Stephenson J Jr, Baker SD, Hidalgo M, Patnaik A, Hammond LA, Weiss G, Goetz A, Siu L, Simmons C, Jolivet J, Rowinsky EK: Troxacitabine, an L-stereoisomeric nucleoside analog, on a five-times-daily schedule: a phase I and pharmacokinetic study in patients with advanced solid malignancies. J Clin Oncol. 2002 Jan 1;20(1):96-109. [11773159 ]
- Lebedev PA: [Selenium deficiency in patients with cardiovascular diseases and its correction with the drug "selena"] Patol Fiziol Eksp Ter. 1996 Jul-Sep;(3):5-7. [8966057 ]
- Shejbal J, Arient M, Kovarikova H: Estimation of deoxycytidine in the urine of humans and laboratory animals. Clin Chim Acta. 1967 May;16(2):324-5. [4962141 ]
- Chen IW, Kereiakes JG, Friedman BI, Saenger EL: Colorimetric analysis of deoxycytidine in urine after separation by ion-exchange column chromatography. Anal Biochem. 1968 May;23(2):230-40. [5657794 ]
- Dientsbier Z, Buric L, Bechyne M, Kovarikova H, Foltynova V, Mostecky H: Deoxypyrimidineuria in the course of chemotherapy in M. Hodgkin. Neoplasma. 1976;23(6):645-65. [1004661 ]
- Dudley E, Lemiere F, Van Dongen W, Langridge JI, El-Sharkawi S, Games DE, Esmans EL, Newton RP: Analysis of urinary nucleosides. III. Identification of 5'-deoxycytidine in urine of a patient with head and neck cancer. Rapid Commun Mass Spectrom. 2003;17(11):1132-6. [12772268 ]
- Arient M, Dienstbier Z, Shejbal J: The post-irradiation excretion of deoxycytidine by urine in man. Minerva Fisiconucl. 1966;10(1):57-60. [5956823 ]
- Tuma P, Samcova E, Kvasnicova V: Improved detection limit for a direct determination of 8-hydroxy-2'-deoxyguanosine in untreated urine samples by capillary electrophoresis with optical detection. J Chromatogr B Analyt Technol Biomed Life Sci. 2004 Dec 25;813(1-2):255-61. [15556541 ]
|
|---|