|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB120066 |
|---|
|
Identification |
|---|
| Name: |
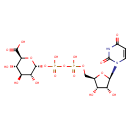
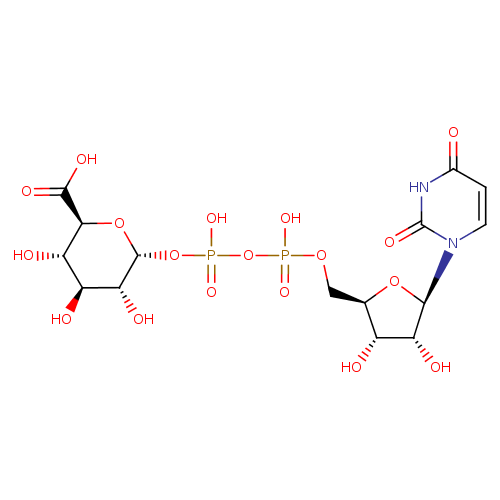
UDP-α-D-glucuronate |
|---|
| Description: | A nucleotide-sugar oxoanion that is a trianion arising from deprotonation of the carboxy and diphosphate OH groups of UDP-α-D-glucuronic acid; major species at pH 7.3. |
|---|
|
Structure |
|
|---|
| Synonyms: | - UDP-α-D-glucuronate
- UDP-α-D-glucuronate trianion
|
|---|
|
Chemical Formula: |
C15H19N2O18P2 |
|---|
| Average Molecular Weight: |
577.265 |
|---|
| Monoisotopic Molecular
Weight: |
580.0343 |
|---|
| InChI Key: |
HDYANYHVCAPMJV-LXQIFKJMSA-K |
|---|
| InChI: | InChI=1S/C15H22N2O18P2/c18-5-1-2-17(15(26)16-5)12-9(22)6(19)4(32-12)3-31-36(27,28)35-37(29,30)34-14-10(23)7(20)8(21)11(33-14)13(24)25/h1-2,4,6-12,14,19-23H,3H2,(H,24,25)(H,27,28)(H,29,30)(H,16,18,26)/p-3/t4-,6-,7+,8+,9-,10-,11+,12-,14-/m1/s1 |
|---|
| CAS
number: |
2616-64-0 |
|---|
| IUPAC Name: | (2S,3S,4S,5R,6R)-6-({[({[(2R,3S,4R,5R)-5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)-3,4,5-trihydroxyoxane-2-carboxylic acid |
|---|
|
Traditional IUPAC Name: |
udp-?-D-glucuronic acid |
|---|
| SMILES: | C3(=CC(NC(N(C1(OC(C(C1O)O)COP(=O)(OP([O-])(OC2(C(C(C(C(O2)C([O-])=O)O)O)O))=O)[O-]))3)=O)=O) |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as pyrimidine nucleotide sugars. These are pyrimidine nucleotides bound to a saccharide derivative through the terminal phosphate group. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Nucleosides, nucleotides, and analogues |
|---|
| Sub Class | Pyrimidine nucleotides |
|---|
|
Direct Parent |
Pyrimidine nucleotide sugars |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Pyrimidine nucleotide sugar
- Pyrimidine ribonucleoside diphosphate
- Pentose phosphate
- Pentose-5-phosphate
- Glucuronic acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Organic pyrophosphate
- Beta-hydroxy acid
- Pyrimidone
- Monoalkyl phosphate
- Hydroxy acid
- Hydropyrimidine
- Monosaccharide
- Pyrimidine
- Organic phosphoric acid derivative
- Alkyl phosphate
- Pyran
- Oxane
- Phosphoric acid ester
- Vinylogous amide
- Oxolane
- Heteroaromatic compound
- Urea
- Secondary alcohol
- Lactam
- Polyol
- Azacycle
- Carboxylic acid derivative
- Carboxylic acid
- Organoheterocyclic compound
- Monocarboxylic acid or derivatives
- Oxacycle
- Alcohol
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organonitrogen compound
- Carbonyl group
- Organooxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aromatic heteromonocyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -3 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Ghosal A, Hapangama N, Yuan Y, Achanfuo-Yeboah J, Iannucci R, Chowdhury S, Alton K, Patrick JE, Zbaida S: Identification of human UDP-glucuronosyltransferase enzyme(s) responsible for the glucuronidation of ezetimibe (Zetia). Drug Metab Dispos. 2004 Mar;32(3):314-20. [14977865 ]
- Quintus J, Kovar KA, Link P, Hamacher H: Urinary excretion of arbutin metabolites after oral administration of bearberry leaf extracts. Planta Med. 2005 Feb;71(2):147-52. [15729623 ]
- Cappiello M, Giuliani L, Rane A, Pacifici GM: Uridine 5'-diphosphoglucuronic acid (UDPGLcUA) in the human fetal liver, kidney and placenta. Eur J Drug Metab Pharmacokinet. 2000 Jul-Dec;25(3-4):161-3. [11420884 ]
- Huskey SW, Doss GA, Miller RR, Schoen WR, Chiu SH: N-glucuronidation reactions. II. Relative N-glucuronidation reactivity of methylbiphenyl tetrazole, methylbiphenyl triazole, and methylbiphenyl imidazole in rat, monkey, and human hepatic microsomes. Drug Metab Dispos. 1994 Jul-Aug;22(4):651-8. [7956743 ]
- Alkharfy KM, Frye RF: High-performance liquid chromatographic assay for acetaminophen glucuronide in human liver microsomes. J Chromatogr B Biomed Sci Appl. 2001 Apr 5;753(2):303-8. [11334344 ]
- Hagenauer B, Salamon A, Thalhammer T, Kunert O, Haslinger E, Klingler P, Senderowicz AM, Sausville EA, Jager W: In vitro glucuronidation of the cyclin-dependent kinase inhibitor flavopiridol by rat and human liver microsomes: involvement of UDP-glucuronosyltransferases 1A1 and 1A9. Drug Metab Dispos. 2001 Apr;29(4 Pt 1):407-14. [11259324 ]
- Cappiello M, Giuliani L, Pacifici GM: Distribution of UDP-glucuronosyltransferase and its endogenous substrate uridine 5'-diphosphoglucuronic acid in human tissues. Eur J Clin Pharmacol. 1991;41(4):345-50. [1804651 ]
- Fondeur-Gelinotte M, Lattard V, Oriol R, Mollicone R, Jacquinet JC, Mulliert G, Gulberti S, Netter P, Magdalou J, Ouzzine M, Fournel-Gigleux S: Phylogenetic and mutational analyses reveal key residues for UDP-glucuronic acid binding and activity of beta1,3-glucuronosyltransferase I (GlcAT-I). Protein Sci. 2006 Jul;15(7):1667-78. [16815917 ]
- Clinical Guide to Laboratory Tests, 2nd Ed. Norbert W. Tietz 1990
|
|---|
| Synthesis Reference: |
Simon, Ethan S.; Grabowski, Sven; Whitesides, George M. Convenient syntheses of cytidine 5'-triphosphate, guanosine 5'-triphosphate, and uridine 5'-triphosphate and their use in the preparation of UDP-glucose, UDP-glucuronic acid, and GDP-mannose. Journal |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|