|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB110846 |
|---|
|
Identification |
|---|
| Name: |
dTTP |
|---|
| Description: | Thymidine triphosphate or TTP is one of the four nucleoside triphosphates that make up DNA. It can be used by DNA ligase to create overlapping "sticky ends" so that protruding ends of opened microbial plasmids maybe closed up. |
|---|
|
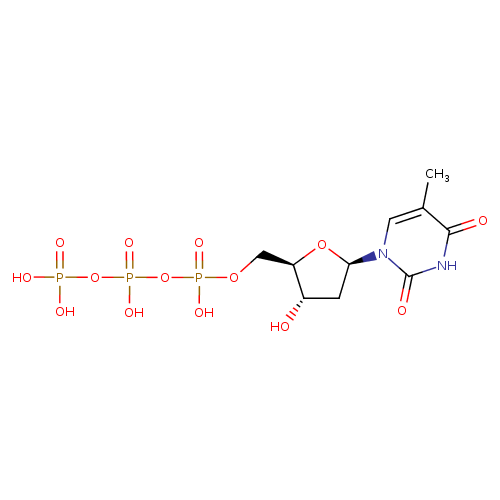
Structure |
|
|---|
| Synonyms: | -
TTP
-
thymidine triphosphate
-
thymidine 5'-triphosphate
|
|---|
|
Chemical Formula: |
C10H13N2O14P3
|
|---|
| Average Molecular Weight: |
478.14 |
|---|
| Monoisotopic Molecular
Weight: |
481.9892627955 |
|---|
| InChI Key: |
NHVNXKFIZYSCEB-XLPZGREQSA-J |
|---|
| InChI: |
InChI=1S/C10H17N2O14P3/c1-5-3-12(10(15)11-9(5)14)8-2-6(13)7(24-8)4-23-28(19,20)26-29(21,22)25-27(16,17)18/h3,6-8,13H,2,4H2,1H3,(H,19,20)(H,21,22)(H,11,14,15)(H2,16,17,18)/p-4/t6-,7+,8+/m0/s1 |
|---|
| CAS
number: |
365-08-2 |
|---|
| IUPAC Name: | thymidine 5'-triphosphate(4−) |
|---|
|
Traditional IUPAC Name: |
dTTP |
|---|
| SMILES: | CC1(=CN(C(=O)NC(=O)1)C2(CC(O)C(COP(=O)([O-])OP(=O)([O-])OP(=O)([O-])[O-])O2)) |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of chemical entities known as pyrimidine 2'-deoxyribonucleoside triphosphates. These are pyrimidine nucleotides with a triphosphate group linked to the ribose moiety lacking a hydroxyl group at position 2. |
|---|
|
Kingdom |
Chemical entities |
|---|
| Super Class | Organic compounds |
|---|
|
Class |
Nucleosides, nucleotides, and analogues |
|---|
| Sub Class | Pyrimidine nucleotides |
|---|
|
Direct Parent |
Pyrimidine 2'-deoxyribonucleoside triphosphates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Pyrimidine 2'-deoxyribonucleoside triphosphate
- Pyrimidone
- Monoalkyl phosphate
- Hydropyrimidine
- Organic phosphoric acid derivative
- Phosphoric acid ester
- Pyrimidine
- Alkyl phosphate
- Oxolane
- Heteroaromatic compound
- Vinylogous amide
- Lactam
- Urea
- Secondary alcohol
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organic oxide
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic oxygen compound
- Alcohol
- Organic nitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aromatic heteromonocyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -4 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
| Spectrum Type | Description | Splash Key | |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-001i-0000900000-d176c8e8a9bdb2d457e5 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-001i-0000900000-d176c8e8a9bdb2d457e5 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-00di-0900000000-970154df2a1ed9a84a2f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | Not Available |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available |
|---|
| 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available |
|---|
|
|---|
|
References |
|---|
| References: |
- Dahlmann N, Ueckermann C: Separation of deoxythymidine-5'-triphosphatase from unspecific hydrolases. A recommended micromethod in the diagnostic evaluation of human carcinoma. Anticancer Res. 1984 Jul-Oct;4(4-5):299-303. [6091528 ]
- Dahlmann N: Human serum thymidine triphosphate nucleotidohydrolase: purification and properties of a new enzyme. Biochemistry. 1982 Dec 21;21(26):6634-9. [6297538 ]
- Schultes BC, Fischbach E, Dahlmann N: Purification and characterization of two different thymidine-5'-triphosphosphate-hydrolysing enzymes in human serum. Biol Chem Hoppe Seyler. 1992 May;373(5):237-47. [1320895 ]
|
|---|
| Synthesis Reference: |
Ikehara, Morio; Ohtsuka, Eiko. Coenzyme analogs. XXI. A new synthesis of thymidine 5'-triphosphate and the use of P1,P2-bis(2-cyanoethyl) pyrophosphate in the nucleoside triphosphate synthesis. Chemical & Pharmaceutical Bulletin (1963), 11(11), 1358-63. |
|---|
| Material Safety Data Sheet (MSDS) |
Download (PDF) |
|---|
|
Links |
|---|
| External Links: |
|
|---|