|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB110025 |
|---|
|
Identification |
|---|
| Name: |
gallate |
|---|
| Description: | A trihydroxybenzoate that is the conjugate base of gallic acid. |
|---|
|
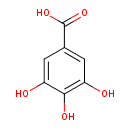
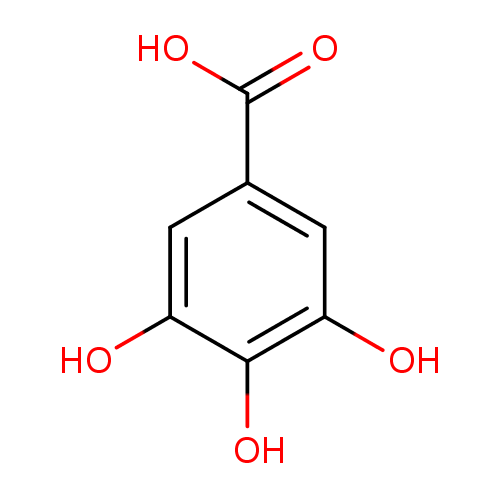
Structure |
|
|---|
| Synonyms: | -
gallic acid
-
3,4,5-trihydroxybenzoic acid
-
3,4,5-trihydroxybenzoate
-
pyrogallol-5-carboxylic acid
-
pyrogallol-5-carboxylate
|
|---|
|
Chemical Formula: |
C7H5O5
|
|---|
| Average Molecular Weight: |
169.11 |
|---|
| Monoisotopic Molecular
Weight: |
170.0215233031 |
|---|
| InChI Key: |
LNTHITQWFMADLM-UHFFFAOYSA-M |
|---|
| InChI: |
InChI=1S/C7H6O5/c8-4-1-3(7(11)12)2-5(9)6(4)10/h1-2,8-10H,(H,11,12)/p-1 |
|---|
| CAS
number: |
149-91-7 |
|---|
| IUPAC Name: | 3,4,5-trihydroxybenzoate |
|---|
|
Traditional IUPAC Name: |
galop |
|---|
| SMILES: | C([O-])(=O)C1(C=C(C(=C(C=1)O)O)O) |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as gallic acid and derivatives. These are compounds containing a 3,4,5-trihydroxybenzoic acid moiety. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
|
Class |
Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
|
Direct Parent |
Gallic acid and derivatives |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Gallic acid or derivatives
- Benzoic acid
- Benzoyl
- Resorcinol
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Phenoxide
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Hydrocarbon derivative
- Organic oxygen compound
- Organic oxide
- Organooxygen compound
- Organic anion
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework |
Aromatic homomonocyclic compounds |
|---|
| External Descriptors |
- trihydroxybenzoate (CHEBI:16918)
- a small molecule (CPD-183)
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
258 - 265 °C |
|---|
| Experimental Properties: |
| Property | Value | Reference |
|---|
| Melting Point | 258 - 265 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | 11.9 mg/mL at 20 °C | Not Available | | LogP | 0.70 | HANSCH,C ET AL. (1995) |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Not Available |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
| Spectrum Type | Description | Splash Key | |
|---|
| GC-MS | GC-MS Spectrum - GC-EI-TOF (Pegasus III TOF-MS system, Leco; GC 6890, Agilent Technologies) | splash10-001i-0791200000-4d3e1baed2effb0bf15b | View in MoNA |
|---|
| GC-MS | GC-MS Spectrum - GC-MS (4 TMS) | splash10-053r-0491700000-68a1d17dd1d0bb6c5354 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS | Not Available |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-0umi-0900000000-e8e6e57cdaf8d518781a | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-0a59-9600000000-8b3c3dc9e263c174a0ac | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-0ue9-9000000000-da4704e31ba4de6b3aca | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - EI-B (HITACHI M-52) , Positive | splash10-0fk9-5900000000-425a258abda96a507ea0 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 10V, Negative | splash10-014i-0900000000-951aa8106825faaca3f9 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 20V, Negative | splash10-004i-0900000000-bdedf4b4b4fd922ec704 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 30V, Negative | splash10-004i-5900000000-46d7a232b169910da462 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 40V, Negative | splash10-004i-9300000000-010429242e38b1b28c13 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-QQ (API3000, Applied Biosystems) 50V, Negative | splash10-0g29-9100000000-fb4dc241b26c248950f7 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | Not Available |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | Not Available |
|---|
| MS | Mass Spectrum (Electron Ionization) | splash10-0fk9-2900000000-1949ca7ce1b8b610c278 | View in MoNA |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available |
|---|
| 1D NMR | 1H NMR Spectrum | Not Available |
|---|
| 1D NMR | 13C NMR Spectrum | Not Available |
|---|
| 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available |
|---|
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Cheng K F; Yip C S; Yeung H W; Kong Y C Leonurine, an improved synthesis. Experientia (1979), 35(5), 571-2. PubMed ID 446644 |
|---|
| Material Safety Data Sheet (MSDS) |
Download (PDF) |
|---|
|
Links |
|---|
| External Links: |
|
|---|