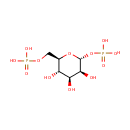|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB004326 |
|---|
|
Identification |
|---|
| Name: |
L-Alanyl-tRNA |
|---|
| Description: | peptidoglycan cross-bridge biosynthesis. The incorporation of. L-alanine required L-alanyl transfer RNA (L-alanyl-tRNA). |
|---|
|
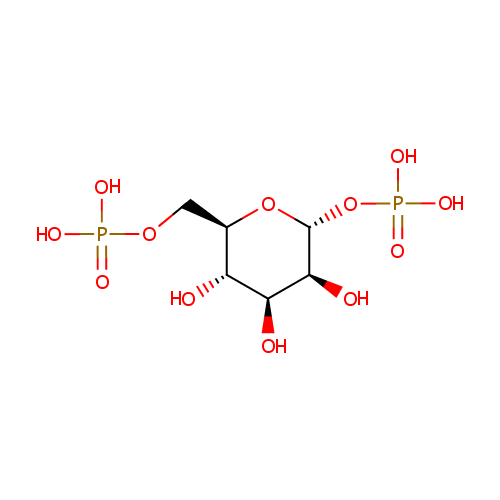
Structure |
|
|---|
| Synonyms: | - Man1,6BP
- Mannose-1,6-bisphosphate
- Mannose-1,6-bisphosphate, (a-D)-isomer
- Mannose-1,6-bisphosphate, (alpha-D)-isomer
- Mannose-1,6-bisphosphate, (L)-isomer
- Mannose-1,6-bisphosphate, (α-D)-isomer
- Mannose-1,6-bisphosphoric acid
- Mannose-1,6-bisphosphoric acid, (a-D)-isomer
- Mannose-1,6-bisphosphoric acid, (alpha-D)-isomer
- Mannose-1,6-bisphosphoric acid, (L)-isomer
- Mannose-1,6-bisphosphoric acid, (α-D)-isomer
|
|---|
|
Chemical Formula: |
C6H14O12P2 |
|---|
| Average Molecular Weight: |
340.114 |
|---|
| Monoisotopic Molecular
Weight: |
339.996049887 |
|---|
| InChI Key: |
RWHOZGRAXYWRNX-RWOPYEJCSA-N |
|---|
| InChI: | InChI=1S/C6H14O12P2/c7-3-2(1-16-19(10,11)12)17-6(5(9)4(3)8)18-20(13,14)15/h2-9H,1H2,(H2,10,11,12)(H2,13,14,15)/t2-,3-,4+,5+,6-/m1/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | {[(2R,3S,4S,5S,6R)-3,4,5-trihydroxy-6-(phosphonooxy)oxan-2-yl]methoxy}phosphonic acid |
|---|
|
Traditional IUPAC Name: |
[(2R,3S,4S,5S,6R)-3,4,5-trihydroxy-6-(phosphonooxy)oxan-2-yl]methoxyphosphonic acid |
|---|
| SMILES: | O[C@H]1[C@H](O)[C@@H](COP(O)(O)=O)O[C@H](OP(O)(O)=O)[C@H]1O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as hexose phosphates. These are carbohydrate derivatives containing a hexose substituted by one or more phosphate groups. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Organooxygen compounds |
|---|
|
Class |
Carbohydrates and carbohydrate conjugates |
|---|
| Sub Class | Monosaccharides |
|---|
|
Direct Parent |
Hexose phosphates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Hexose phosphate
- Monosaccharide phosphate
- Monoalkyl phosphate
- Alkyl phosphate
- Phosphoric acid ester
- Oxane
- Organic phosphoric acid derivative
- Organic phosphate
- Secondary alcohol
- 1,2-diol
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Organic oxide
- Hydrocarbon derivative
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -4 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | 3036654 | | Kegg ID | C00886 | | ChemSpider ID | 2300632 | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|