|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB003592 |
|---|
|
Identification |
|---|
| Name: |
UDP-beta-L-Threo-pentapyranos-4-ulose |
|---|
| Description: | UDP-beta-L-Threo-pentapyranos-4-ulose is an intermediate in the polymixin resistance pathway. It is a substrate for the enzyme UDP-4-amino-4-deoxy-L-arabinose aminotransferase which catalyzes the reaction UDP-4-amino-4-deoxy-beta-L-arabinopyranose + 2-oxoglutarate = UDP-beta-L-threo-pentapyranos-4-ulose + L-glutamate. Some Gram-negative bacteria, specifically Salmonella typhimurium and Pseudomonas aeruginosa, can become resistant to polymyxin by the modification of their lipid A structure via the attachment of 4-amino-4-deoxy-L-arabinopyranose (L-Ara4N) groups to one or more phosphate groups. This addition causes an absolute increase in lipid A charge, thus lowering the affinity of positively charged polymyxins. |
|---|
|
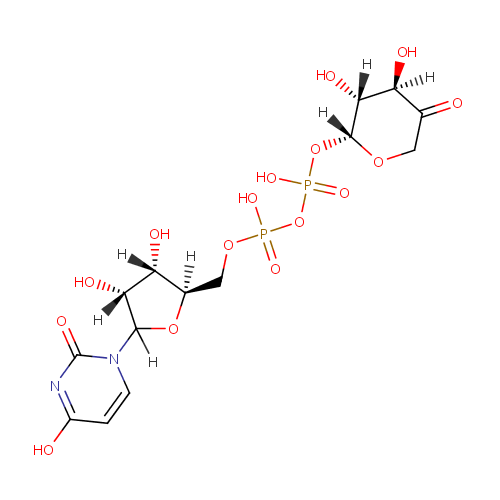
Structure |
|
|---|
| Synonyms: | - UDP-b-L-threo-Pentapyranos-4-ulose
- UDP-β-L-threo-Pentapyranos-4-ulose
|
|---|
|
Chemical Formula: |
C14H20N2O16P2 |
|---|
| Average Molecular Weight: |
534.2599 |
|---|
| Monoisotopic Molecular
Weight: |
534.028805626 |
|---|
| InChI Key: |
URJZIQLTPCJVMW-SVROINDSSA-N |
|---|
| InChI: | InChI=1S/C14H20N2O16P2/c17-5-3-28-13(11(22)8(5)19)31-34(26,27)32-33(24,25)29-4-6-9(20)10(21)12(30-6)16-2-1-7(18)15-14(16)23/h1-2,6,8-13,19-22H,3-4H2,(H,24,25)(H,26,27)(H,15,18,23)/t6-,8+,9-,10-,11-,12?,13-/m1/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | {[(2R,3S,4R)-3,4-dihydroxy-5-(4-hydroxy-2-oxo-1,2-dihydropyrimidin-1-yl)oxolan-2-yl]methoxy}[({[(2R,3R,4R)-3,4-dihydroxy-5-oxooxan-2-yl]oxy}(hydroxy)phosphoryl)oxy]phosphinic acid |
|---|
|
Traditional IUPAC Name: |
[(2R,3S,4R)-3,4-dihydroxy-5-(4-hydroxy-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy({[(2R,3R,4R)-3,4-dihydroxy-5-oxooxan-2-yl]oxy(hydroxy)phosphoryl}oxy)phosphinic acid |
|---|
| SMILES: | [H][C@]1(COP(O)(=O)OP(O)(=O)O[C@@]2([H])OCC(=O)[C@]([H])(O)[C@@]2([H])O)OC([H])(N2C=CC(O)=NC2=O)[C@]([H])(O)[C@]1([H])O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as pyrimidine ribonucleoside diphosphates. These are pyrimidine ribobucleotides with diphosphate group linked to the ribose moiety. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
|
Class |
Pyrimidine nucleotides |
|---|
| Sub Class | Pyrimidine ribonucleotides |
|---|
|
Direct Parent |
Pyrimidine ribonucleoside diphosphates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Pyrimidine ribonucleoside diphosphate
- N-glycosyl compound
- Glycosyl compound
- Organic pyrophosphate
- Monosaccharide phosphate
- Hydroxypyrimidine
- Monoalkyl phosphate
- Pyrimidone
- Alkyl phosphate
- Pyrimidine
- Phosphoric acid ester
- Oxane
- Organic phosphoric acid derivative
- Organic phosphate
- Monosaccharide
- Hydropyrimidine
- Heteroaromatic compound
- Oxolane
- Cyclic ketone
- Secondary alcohol
- Ketone
- 1,2-diol
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Alcohol
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework |
Aromatic heteromonocyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -2 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
Not Available |
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
Not Available |
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | Not Available | | HMDB ID | Not Available | | Pubchem Compound ID | 25244749 | | Kegg ID | Not Available | | ChemSpider ID | Not Available | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|