|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB002048 |
|---|
|
Identification |
|---|
| Name: |
Phosphatidylglycerophosphate |
|---|
| Description: | Phosphatidylglycerophosphate belongs to the class of Phosphatidylglycerophosphates. These are glycerophosphoglycerophosphates in which two fatty acids are bonded to the 1-glycerol moiety through ester linkages. |
|---|
|
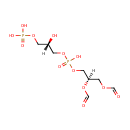
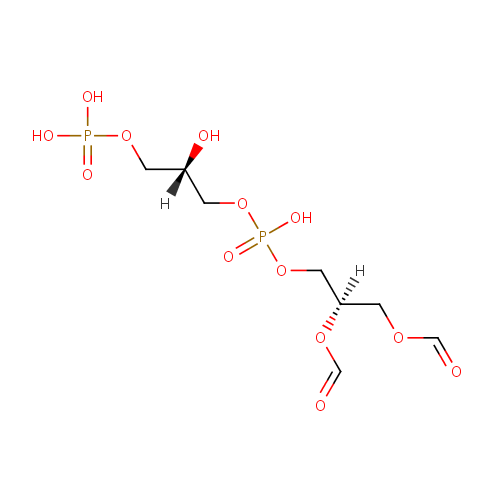
Structure |
|
|---|
| Synonyms: | - 1,2-Diacyl-sn-glycero-3-phospho-sn-glycerol 3'-phosphate
- 1,2-Diacyl-sn-glycero-3-phospho-sn-glycerol 3'-phosphoric acid
- 3(3-Phosphatidyl-)L-glycerol 1-phosphate
- 3(3-Phosphatidyl-)L-glycerol 1-phosphoric acid
- 3(3-Sn-Phosphatidyl)-sn-glycerol 1-phosphate
- 3(3-sn-Phosphatidyl)-sn-glycerol 1-phosphoric acid
- Phosphatidylglycerol phosphates
- Phosphatidylglycerophosphate
- Phosphatidylglycerophosphoric acid
|
|---|
|
Chemical Formula: |
C8H16O13P2 |
|---|
| Average Molecular Weight: |
382.1524 |
|---|
| Monoisotopic Molecular
Weight: |
382.006613622 |
|---|
| InChI Key: |
ZSPJCYNIVYENCI-JGVFFNPUSA-N |
|---|
| InChI: | InChI=1S/C8H16O13P2/c9-5-17-3-8(18-6-10)4-21-23(15,16)20-2-7(11)1-19-22(12,13)14/h5-8,11H,1-4H2,(H,15,16)(H2,12,13,14)/t7-,8+/m0/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | [(2S)-3-({[(2R)-2,3-bis(formyloxy)propoxy](hydroxy)phosphoryl}oxy)-2-hydroxypropoxy]phosphonic acid |
|---|
|
Traditional IUPAC Name: |
(2S)-3-{[(2R)-2,3-bis(formyloxy)propoxy(hydroxy)phosphoryl]oxy}-2-hydroxypropoxyphosphonic acid |
|---|
| SMILES: | [H][C@](O)(COP(O)(O)=O)COP(O)(=O)OC[C@@]([H])(COC=O)OC=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as phosphatidylglycerophosphates. These are glycerophosphoglycerophosphates in which two fatty acids are bonded to the 1-glycerol moiety through ester linkages. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Glycerophospholipids |
|---|
| Sub Class | Glycerophosphoglycerophosphates |
|---|
|
Direct Parent |
Phosphatidylglycerophosphates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Diacylglycerophosphoglycerophosphate
- Sn-glycerol-3-phosphate
- Dialkyl phosphate
- Monoalkyl phosphate
- Alkyl phosphate
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Organic phosphate
- Secondary alcohol
- Carboxylic acid ester
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -3 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- Yurtsever D. (2007). Fatty acid methyl ester profiling of Enterococcus and Esherichia coli for microbial source tracking. M.sc. Thesis. Villanova University: U.S.A
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
| Resource | Link |
|---|
| CHEBI ID | 17264 | | HMDB ID | Not Available | | Pubchem Compound ID | 46873832 | | Kegg ID | C03892 | | ChemSpider ID | Not Available | | Wikipedia ID | Not Available | | BioCyc ID | Not Available |
|
|---|