|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB001409 |
|---|
|
Identification |
|---|
| Name: |
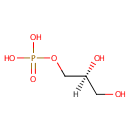
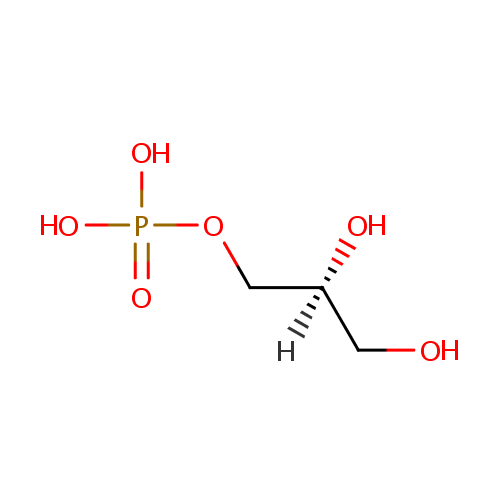
Sn-Glycerol-1-phosphate |
|---|
| Description: | Sn-glycerol-1-phosphate is a glycerol phosphate. It is a key substrate in phospholipid synthesis and phosphatidylglycerol sbiosynthesis. The sn-glycerol-3-phosphate acyltransferase (plsB) of Pseudomonas aeruginosa is a key regulatory enzyme that catalyzes the first committed step in phospholipid biosynthesis. (PMID 9393688) Fosmidomycin is a hydrophilic molecule and in many bacteria its uptake is an active process involving a cAMP dependent glycerol-3-phosphate transporter (GlpT). (PMID 18489786) |
|---|
|
Structure |
|
|---|
| Synonyms: | - .alpha.-glycerophosphate
- .alpha.-glycerophosphorate
- .alpha.-glycerophosphoric acid
- 1,2,3-Propanetriol, 1-(dihydrogen phosphate)
- 1,2,3-Propanetriol, 1-(dihydrogen phosphoric acid)
- 1-Glycerophosphate
- 1-Glycerophosphorate
- 1-Glycerophosphoric acid
- 2,3-Dihydroxypropyl dihydrogen phosphate
- 2,3-Dihydroxypropyl dihydrogen phosphoric acid
- 2,3-Hydroxy-1-propyl dihydrogen phosphate
- 2,3-Hydroxy-1-propyl dihydrogen phosphoric acid
- 3-Glycerophosphate
- 3-Glycerophosphoric acid
- 3-Phosphoglycerol
- a-Glycerophosphate
- a-Glycerophosphorate
- a-Glycerophosphoric acid
- a-Phosphoglycerol
- alpha-Glycerophosphate
- Alpha-Glycerophosphorate
- Alpha-Glycerophosphoric acid
- Alpha-Phosphoglycerol
- BBR
- D-Glycerol 3-phosphate
- D-Glycerol 3-phosphoric acid
- Glycerol .alpha.-phosphate
- Glycerol .alpha.-phosphoric acid
- Glycerol 1-(dihydrogen phosphate)
- Glycerol 1-(dihydrogen phosphoric acid)
- Glycerol 1-phosphate
- Glycerol 1-phosphoric acid
- Glycerol 3-phosphate
- Glycerol 3-phosphoric acid
- Glycerol a-phosphate
- Glycerol a-phosphoric acid
- Glycerol alpha-phosphate
- Glycerol alpha-phosphoric acid
- Glycerol dihydrogen phosphate
- Glycerol dihydrogen phosphoric acid
- Glycerol α-phosphate
- Glycerol α-phosphoric acid
- Glycerol, 1-(dihydrogen phosphate)
- Glycerol, 1-(dihydrogen phosphate) (8CI)
- Glycerol, 1-(dihydrogen phosphoric acid)
- Glycerol, 1-(dihydrogen phosphoric acid) (8ci)
- GLYCEROL-3-PHOSPHATE
- GLYCEROL-3-phosphoric acid
- Glycerophosphate
- Glycerophosphate i
- Glycerophosphorate
- Glycerophosphorate I
- Glycerophosphoric acid
- Glycerophosphoric acid I
- L-Glycerol 1-phosphate
- L-Glycerol 1-phosphoric acid
- Sn-Glycerol 1-phosphate
- sn-Glycerol 1-phosphoric acid
- sn-Glycerol-1-phosphoric acid
- SN-glyceryl phosphate
- sn-Glyceryl phosphoric acid
- [(2S)-2,3-dihydroxypropyl] dihydrogen phosphate
- [(2S)-2,3-Dihydroxypropyl] dihydrogen phosphoric acid
- α-Glycerophosphate
- α-Glycerophosphorate
- α-Glycerophosphoric acid
- α-Phosphoglycerol
|
|---|
|
Chemical Formula: |
C3H9O6P |
|---|
| Average Molecular Weight: |
172.0737 |
|---|
| Monoisotopic Molecular
Weight: |
172.013674532 |
|---|
| InChI Key: |
AWUCVROLDVIAJX-VKHMYHEASA-N |
|---|
| InChI: | InChI=1S/C3H9O6P/c4-1-3(5)2-9-10(6,7)8/h3-5H,1-2H2,(H2,6,7,8)/t3-/m0/s1 |
|---|
| CAS
number: |
5746-57-6 |
|---|
| IUPAC Name: | [(2S)-2,3-dihydroxypropoxy]phosphonic acid |
|---|
|
Traditional IUPAC Name: |
glycerophosphate |
|---|
| SMILES: | [H][C@](O)(CO)COP(O)(O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as glycerophosphates. These are compounds containing a glycerol linked to a phosphate group. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Glycerophospholipids |
|---|
| Sub Class | Glycerophosphates |
|---|
|
Direct Parent |
Glycerophosphates |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Sn-glycerol-3-phosphate
- Monoalkyl phosphate
- Alkyl phosphate
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Organic phosphate
- Saccharide
- Secondary alcohol
- 1,2-diol
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -2 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
Not Available |
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Brown, A. C., Parish, T. (2008). "Dxr is essential in Mycobacterium tuberculosis and fosmidomycin resistance is due to a lack of uptake." BMC Microbiol 8:78. Pubmed: 18489786
- Heath, R. J., Goldfine, H., Rock, C. O. (1997). "A gene (plsD) from Clostridium butyricum that functionally substitutes for the sn-glycerol-3-phosphate acyltransferase gene (plsB) of Escherichia coli." J Bacteriol 179:7257-7263. Pubmed: 9393688
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|