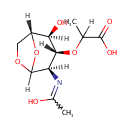
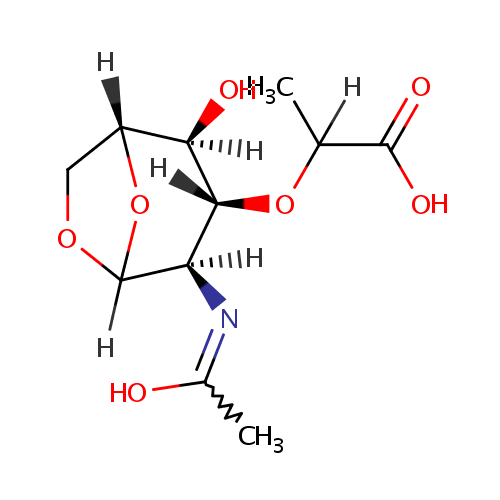
1,6-Anhydro-N-acetylmuramate (PAMDB001327)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB001327 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | 1,6-Anhydro-N-acetylmuramate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | 1,6-anhMurNAC is an Amino Sugar. These are compounds having one alcoholic hydroxy group replaced by an amino group; systematically known as x-amino-x-deoxymonosaccharides. 1,6-anhydro-N-acetylmuramic acid (anhMurNAc) is one of the components of murein that is recycled inside the cell. Although exogenously provided anhMurNAc can be taken up by Pseudomonas aeruginosa, it can not serve as the sole source of carbon for growth. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C11H17NO7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 275.2552 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 275.100501903 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | ZFEGYUMHFZOYIY-NFSFVEDMSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C11H17NO7/c1-4(10(15)16)18-9-7(12-5(2)13)11-17-3-6(19-11)8(9)14/h4,6-9,11,14H,3H2,1-2H3,(H,12,13)(H,15,16)/t4?,6-,7-,8-,9-,11?/m1/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | 2-{[(1R,2S,3R,4R)-2-hydroxy-4-[(1-hydroxyethylidene)amino]-6,8-dioxabicyclo[3.2.1]octan-3-yl]oxy}propanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | 2-{[(1R,2S,3R,4R)-2-hydroxy-4-[(1-hydroxyethylidene)amino]-6,8-dioxabicyclo[3.2.1]octan-3-yl]oxy}propanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | [H]C(C)(O[C@@]1([H])[C@]([H])(O)[C@@]2([H])COC([H])(O2)[C@]1([H])N=C(C)O)C(O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as oxepanes. These are compounds containing an oxepane ring, which is a seven-member saturated aliphatic heterocycle with one oxygen and six carbon atoms. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organoheterocyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Oxepanes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Oxepanes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic heteropolycyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | 1,6-Anhydrous-N-Acetylmuramyl-tetrapeptide + Water > L-Alanine-D-glutamate-meso-2,6-diaminoheptanedioate-D-alanine + 1,6-Anhydro-N-acetylmuramate 1,6-Anhydrous-N-Acetylmuramyl-tripeptide + Water > L-alanine-D-glutamate-meso-2,6-diaminoheptanedioate + 1,6-Anhydro-N-acetylmuramate N-Acetyl-D-glucosamine(anhydrous)N-Acetylmuramic acid + Water > N-Acetyl-D-glucosamine + 1,6-Anhydro-N-acetylmuramate 1,6-Anhydro-N-acetylmuramate + Adenosine triphosphate + Water > Hydrogen ion + N-Acetylmuramic acid 6-phosphate + ADP <i>N</i>-acetyl-β-D-glucosamine(anhydrous)-<i>N</i>-acetylmuramate + Water N-Acetyl-D-glucosamine + 1,6-Anhydro-N-acetylmuramate Adenosine triphosphate + Water + 1,6-Anhydro-N-acetylmuramate <> Adenosine diphosphate + MurNAc-6-P + ADP | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||