|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB001122 |
|---|
|
Identification |
|---|
| Name: |
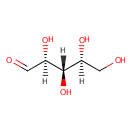
L-Ribose |
|---|
| Description: | Ribose is an organic compound that occurs widely in nature. It is an aldopentose, a monosaccharide containing five carbon atoms that in its acyclic form has an aldehyde functional group at one end. Typically, ribose exists in the cyclic form. It comprises the backbone of RNA, a biopolymer that is the basis of genetic transcription. It is related to deoxyribose, as found in DNA, by the removal of one hydroxy group. Once phosphorylated, ribose can become a subunit of ATP, NADH, and several other compounds that are critical to metabolism. Ribose exists in two enantiomeric forms, primarily as D-ribose. L-ribose is the synthetic mirror image of D-ribose. (Wikipedia) In Pseudomonas aeruginosa, L-ribose can act as an competitive inhibitor of glucose dehydrogenase. (EcoCyc) |
|---|
|
Structure |
|
|---|
| Synonyms: | - (2S,3S,4S)-2,3,4,5-tetrahydroxypentanal
- Aldehydo-L-ribo-pentose
- Aldehydo-L-ribose
- Arabinose
|
|---|
|
Chemical Formula: |
C5H10O5 |
|---|
| Average Molecular Weight: |
150.1299 |
|---|
| Monoisotopic Molecular
Weight: |
150.05282343 |
|---|
| InChI Key: |
PYMYPHUHKUWMLA-MROZADKFSA-N |
|---|
| InChI: | InChI=1S/C5H10O5/c6-1-3(8)5(10)4(9)2-7/h1,3-5,7-10H,2H2/t3-,4+,5-/m1/s1 |
|---|
| CAS
number: |
24259-59-4 |
|---|
| IUPAC Name: | (2S,3S,4S)-2,3,4,5-tetrahydroxypentanal |
|---|
|
Traditional IUPAC Name: |
L-ribose |
|---|
| SMILES: | [H][C@](O)(CO)[C@]([H])(O)[C@]([H])(O)C=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as monosaccharides. These are compounds containing one carbohydrate unit not glycosidically linked to another such unit, and no set of two or more glycosidically linked carbohydrate units. Monosaccharides have the general formula CnH2nOn. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Organooxygen compounds |
|---|
|
Class |
Carbohydrates and carbohydrate conjugates |
|---|
| Sub Class | Monosaccharides |
|---|
|
Direct Parent |
Monosaccharides |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Monosaccharide
- Beta-hydroxy aldehyde
- Alpha-hydroxyaldehyde
- Secondary alcohol
- Polyol
- 1,2-diol
- Hydrocarbon derivative
- Short-chain aldehyde
- Primary alcohol
- Carbonyl group
- Aldehyde
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | 0 |
|---|
|
Melting point: |
99 ?C, 372 K, 210 ?F |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
Not Available |
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|