|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB001040 |
|---|
|
Identification |
|---|
| Name: |
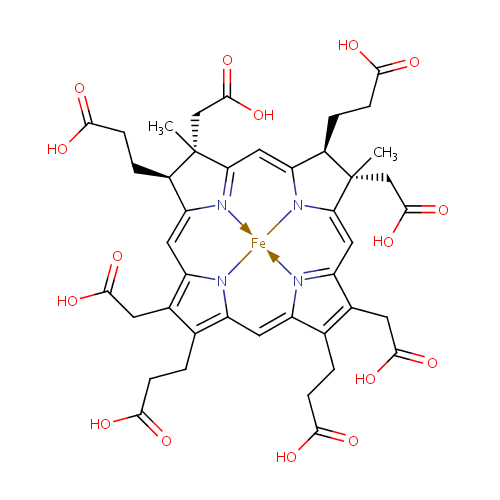
Siroheme |
|---|
| Description: | Siroheme belongs to the class of Precorrins. These are intermediates formed by methylation at one or more of the four rings prior to the formation of the macrocyclic corrin ring. (inferred from compound structure)Siroheme (or sirohaem) is a heme-like prosthetic group used by some enzymes to accomplish the six-electron reduction of sulfur and nitrogen. Siroheme is synthesized from uroporphyrinogen III, a heme and vitamin B12 precursor. It plays a major role in the sulfur assimilation pathway: converting sulfite to a biologically useful sulfide, which can be incorporated into the organic compound homocysteine. (WikiPedia) |
|---|
|
Structure |
|
|---|
| Synonyms: | - Iron(2+) (2S,3S,7S,8S)-2,7,13,17-tetrakis(2-carboxyethyl)-3,8,12,18-tetrakis(carboxymethyl)-3,8-dimethyl-7,8-dihydro-2H,3H-porphine-21,23-diide
- Iron(2+); 3-(2S,3S,7S,8S)-7,13,17-tris(2-carboxyethyl)-3,8,12,18-tetrakis(carboxymethyl)-3,8-dimethyl-2,7-dihydroporphyrin-21,23-diid-2-ylpropanoate
- Iron(2+); 3-(2S,3S,7S,8S)-7,13,17-tris(2-carboxyethyl)-3,8,12,18-tetrakis(carboxymethyl)-3,8-dimethyl-2,7-dihydroporphyrin-21,23-diid-2-ylpropanoic acid
|
|---|
|
Chemical Formula: |
C42H44FeN4O16 |
|---|
| Average Molecular Weight: |
916.661 |
|---|
| Monoisotopic Molecular
Weight: |
916.210173513 |
|---|
| InChI Key: |
DLKSSIHHLYNIKN-QIISWYHFSA-L |
|---|
| InChI: | InChI=1S/C42H46N4O16.Fe/c1-41(17-39(59)60)23(5-9-35(51)52)29-14-27-21(11-37(55)56)19(3-7-33(47)48)25(43-27)13-26-20(4-8-34(49)50)22(12-38(57)58)28(44-26)15-31-42(2,18-40(61)62)24(6-10-36(53)54)30(46-31)16-32(41)45-29;/h13-16,23-24H,3-12,17-18H2,1-2H3,(H10,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62);/q;+2/p-2/t23-,24-,41+,42+;/m1./s1 |
|---|
| CAS
number: |
52553-42-1 |
|---|
| IUPAC Name: | 3-[(4S,5S,9S,10S,11Z,16Z)-9,15,19-tris(2-carboxyethyl)-5,10,14,20-tetrakis(carboxymethyl)-5,10-dimethyl-21,23,24,25-tetraaza-22-ferrahexacyclo[9.9.3.1?,??1??,???0????.0?????]pentacosa-1(20),2,6(25),7,11,13(24),14,16,18-nonaen-4-yl]propanoic acid |
|---|
|
Traditional IUPAC Name: |
3-[(4S,5S,9S,10S,11Z,16Z)-9,15,19-tris(2-carboxyethyl)-5,10,14,20-tetrakis(carboxymethyl)-5,10-dimethyl-21,23,24,25-tetraaza-22-ferrahexacyclo[9.9.3.1?,??1??,???0????.0?????]pentacosa-1(20),2,6(25),7,11,13(24),14,16,18-nonaen-4-yl]propanoic acid |
|---|
| SMILES: | C[C@]1(CC(O)=O)[C@H](CCC(O)=O)\C2=C\C3=C(CC(O)=O)C(CCC(O)=O)=C4\C=C5/N=C(/C=C6\N([Fe]N34)\C(=C/C1=N2)[C@@H](CCC(O)=O)[C@]6(C)CC(O)=O)C(CC(O)=O)=C5CCC(O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as metallotetrapyrroles. These are polycyclic compounds containing a tetrapyrrole skeleton combined with a metal atom. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
|
Class |
Tetrapyrroles and derivatives |
|---|
| Sub Class | Metallotetrapyrroles |
|---|
|
Direct Parent |
Metallotetrapyrroles |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Metallotetrapyrrole skeleton
- Substituted pyrrole
- Heteroaromatic compound
- Pyrroline
- Pyrrolidine
- Pyrrole
- Ketimine
- Azacycle
- Organic metal salt
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Metalloheterocycle
- Carboxylic acid
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organic transition metal salt
- Organooxygen compound
- Organonitrogen compound
- Imine
- Carbonyl group
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework |
Aromatic heteropolycyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -8 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- van der Werf, M. J., Overkamp, K. M., Muilwijk, B., Coulier, L., Hankemeier, T. (2007). "Microbial metabolomics: toward a platform with full metabolome coverage." Anal Biochem 370:17-25. Pubmed: 17765195
- Winder, C. L., Dunn, W. B., Schuler, S., Broadhurst, D., Jarvis, R., Stephens, G. M., Goodacre, R. (2008). "Global metabolic profiling of Escherichia coli cultures: an evaluation of methods for quenching and extraction of intracellular metabolites." Anal Chem 80:2939-2948. Pubmed: 18331064
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|