|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB000968 |
|---|
|
Identification |
|---|
| Name: |
Benzoyl acetyl-CoA |
|---|
| Description: | Benzoyl acetyl-CoA is a member of the chemical class known as Coenzyme A and Derivatives. These are derivative of vitamin B5 containing a 4'-phosphopantetheine moiety attached to a diphospho-adenosine. |
|---|
|
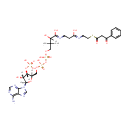
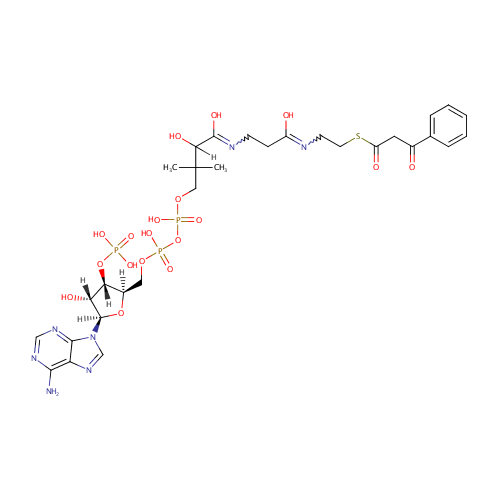
Structure |
|
|---|
| Synonyms: | - p-hydroxybenzoyl-acetyl-CoA
- Benzoyl acetyl coenzyme a
- p-Hydroxybenzoyl-acetyl-CoA
|
|---|
|
Chemical Formula: |
C30H42N7O18P3S |
|---|
| Average Molecular Weight: |
913.677 |
|---|
| Monoisotopic Molecular
Weight: |
913.151987801 |
|---|
| InChI Key: |
NHDPIYICCBKNNJ-YUXRTGJASA-N |
|---|
| InChI: | InChI=1S/C30H42N7O18P3S/c1-30(2,25(42)28(43)33-9-8-20(39)32-10-11-59-21(40)12-18(38)17-6-4-3-5-7-17)14-52-58(49,50)55-57(47,48)51-13-19-24(54-56(44,45)46)23(41)29(53-19)37-16-36-22-26(31)34-15-35-27(22)37/h3-7,15-16,19,23-25,29,41-42H,8-14H2,1-2H3,(H,32,39)(H,33,43)(H,47,48)(H,49,50)(H2,31,34,35)(H2,44,45,46)/t19-,23-,24-,25?,29-/m1/s1 |
|---|
| CAS
number: |
Not Available |
|---|
| IUPAC Name: | 4-({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)oxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)-2-hydroxy-3,3-dimethyl-N-[2-({2-[(3-oxo-3-phenylpropanoyl)sulfanyl]ethyl}-C-hydroxycarbonimidoyl)ethyl]butanimidic acid |
|---|
|
Traditional IUPAC Name: |
benzoyl acetyl-coa |
|---|
| SMILES: | [H]C(O)(C(O)=NCCC(O)=NCCSC(=O)CC(=O)C1=CC=CC=C1)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@@]1([H])O[C@@]([H])(N2C=NC3=C(N)N=CN=C23)[C@]([H])(O)[C@]1([H])OP(O)(O)=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as 3-oxo-acyl coas. These are organic compounds containing a 3-oxo acylated coenzyme A derivative. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Fatty Acyls |
|---|
| Sub Class | Fatty acyl thioesters |
|---|
|
Direct Parent |
3-oxo-acyl CoAs |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Coenzyme a or derivatives
- Purine ribonucleoside diphosphate
- Purine ribonucleoside 3',5'-bisphosphate
- N-glycosyl compound
- Glycosyl compound
- Organic pyrophosphate
- Monosaccharide phosphate
- 6-aminopurine
- Purine
- Imidazopyrimidine
- Acetophenone
- Aryl alkyl ketone
- Aryl ketone
- Benzoyl
- Monoalkyl phosphate
- Aminopyrimidine
- Imidolactam
- Benzenoid
- Alkyl phosphate
- 1,3-dicarbonyl compound
- Pyrimidine
- Primary aromatic amine
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Organic phosphate
- N-substituted imidazole
- Monosaccharide
- Saccharide
- Monocyclic benzene moiety
- Heteroaromatic compound
- Oxolane
- Imidazole
- Azole
- Thiocarboxylic acid ester
- Secondary alcohol
- Ketone
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Sulfenyl compound
- Thioether
- Thiocarboxylic acid or derivatives
- Carboxylic acid derivative
- Carboximidic acid derivative
- Carboximidic acid
- Hydrocarbon derivative
- Primary amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Amine
- Alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework |
Aromatic heteropolycyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Not Available |
|---|
| Charge: | -3 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
- Ethylbenzene degradation pae00642
- Microbial metabolism in diverse environments pae01120
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|