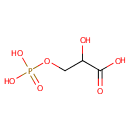
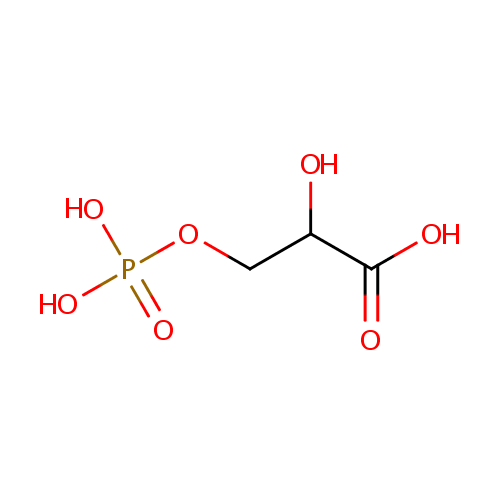
3-Phosphoglycerate (PAMDB000628)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000628 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name: | 3-Phosphoglycerate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | 3-phosphoglyceric acid (3PG) is a 3-carbon molecule that is a metabolic intermediate in both glycolysis and the Calvin cycle. This chemical is often termed PGA when referring to the Calvin cycle. In the Calvin cycle, two glycerate 3-phosphate molecules are reduced to form two molecules of glyceraldehyde 3-phosphate (GALP). (wikipedia) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C3H7O7P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 186.0572 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 185.99293909 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | OSJPPGNTCRNQQC-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C3H7O7P/c4-2(3(5)6)1-10-11(7,8)9/h2,4H,1H2,(H,5,6)(H2,7,8,9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 820-11-1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | 2-hydroxy-3-(phosphonooxy)propanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | phosphoglycerate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | OC(COP(O)(O)=O)C(O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as sugar acids and derivatives. These are compounds containing a saccharide unit which bears a carboxylic acid group. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organooxygen compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carbohydrates and carbohydrate conjugates | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Sugar acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Sugar acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | 2-Phospho-D-glyceric acid <> 3-Phosphoglycerate Adenosine triphosphate + Glyceric acid > 3-Phosphoglycerate + ADP + Hydrogen ion 3-Phosphoglycerate + NAD > Phosphohydroxypyruvic acid + Hydrogen ion + NADH 3-Phosphoglycerate + Adenosine triphosphate <> Glyceric acid 1,3-biphosphate + ADP Glyceric acid 1,3-biphosphate + Adenosine diphosphate + Glyceric acid 1,3-biphosphate + ADP > Adenosine triphosphate + 3-Phosphoglyceric acid + 3-Phosphoglycerate 3-Phosphoglyceric acid + Adenosine triphosphate + 3-Phosphoglycerate > 3-phospho-D-glyceroyl phosphate + Adenosine diphosphate + ADP 3-Phosphoglyceric acid + 3-Phosphoglycerate > 2-Phospho-D-glyceric acid 2-Phosphoglyceric acid + 2-Phosphoglyceric acid > 3-Phosphoglyceric acid + 3-Phosphoglycerate Glyceric acid + Adenosine triphosphate > Adenosine diphosphate + Hydrogen ion + 3-Phosphoglyceric acid + ADP + 3-Phosphoglycerate 3-Phosphoglyceric acid + NAD + 3-Phosphoglycerate > NADH + Hydrogen ion + Phosphohydroxypyruvic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Jakoby, Wm. B.; Brummond, Dewey O.; Ochoa, Severo. Formation of 3-phosphoglyceric acid by carbon dioxide fixation with spinach-leaf enzymes. Journal of Biological Chemistry (1956), 218 811-22. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Download (PDF) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||