|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB000605 |
|---|
|
Identification |
|---|
| Name: |
2-Keto-3-methyl-valerate |
|---|
| Description: | 3-Methyl-2-oxovaleric acid is a metabolite of isoleucine in man, animals and bacteria. It is the alpha-keto acid analogue of isoleucine. 3-Methyl-2-oxovaleric acid is produced from isoleucine by cytosolic branched chain aminotransferase 1 (EC:2.6.1.42), whereupon it is further degraded by branched chain keto acid dehydrogenase E1 to 2-Methyl-1-hydroxybutyl-ThPP. |
|---|
|
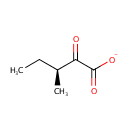
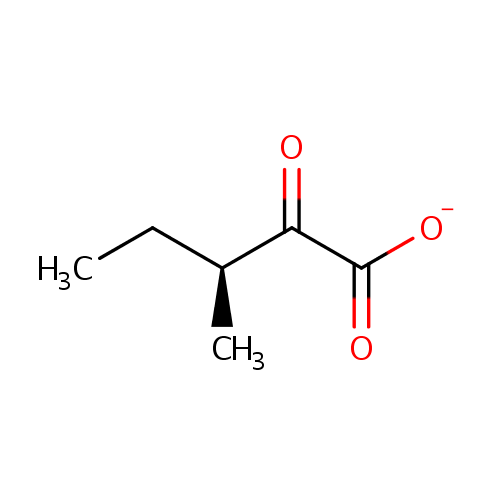
Structure |
|
|---|
| Synonyms: | - α-keto-β-methyl-valerate
- α-keto-β-methyl-valeric acid
- α-keto-methylvalerate
- α-keto-methylvaleric acid
- (3S)-3-Methyl-2-oxopentanoate
- (3R)-3-Methyl-2-oxopentanoate
- (3R)-3-Methyl-2-oxopentanoic acid
- (3S)-3-methyl-2-oxopentanoate
- (3S)-3-methyl-2-oxopentanoic acid
- (S)-2-oxo-3-methylpentanoate
- (S)-3-methyl-2-oxopentanoate
- (R)-3-methyl-2-oxopentanoate
- (R)-3-methyl-2-oxopentanoic acid
- (S)-2-oxo-3-methylpentanoate
- (S)-2-oxo-3-methylpentanoic acid
- (S)-3-methyl-2-oxopentanoate
- (S)-3-methyl-2-oxopentanoic acid
- (S)-3-methyl-2-oxovalerate
- (S)-3-methyl-2-oxovaleric acid
- 2-keto-3-methyl-valerate
- 2-Keto-3-methyl-valeric acid
- 2-keto-3-methylvalerate
- 2-keto-3-methylvaleric acid
- 2-Oxo-3-methyl-N-valerate
- 2-Oxo-3-methyl-N-valeric acid
- 2-Oxo-3-methylpentanoate
- 2-Oxo-3-methylpentanoic acid
- 2-Oxo-3-methylvalerate
- 2-Oxo-3-methylvaleric acid
- 2-Oxoisoleucine
- 2-Oxokolavenate
- 2-Oxokolavenic acid
- 3-Methyl-2-oxo-pentanoate
- 3-Methyl-2-oxo-pentanoic acid
- 3-Methyl-2-oxo-Valerate
- 3-Methyl-2-oxo-Valeric acid
- 3-Methyl-2-oxopentanoate
- 3-Methyl-2-oxopentanoic acid
- 3-Methyl-2-oxovalerate
- 3-Methyl-2-oxovaleric
- 3-Methyl-2-oxovaleric acid
- A-Keto-b-methyl-N-valerate
- A-Keto-b-methyl-N-valeric acid
- a-keto-b-Methyl-valerate
- a-keto-b-Methyl-valeric acid
- A-Keto-b-methylvalerate
- A-Keto-b-methylvaleric acid
- a-keto-Methylvalerate
- a-keto-Methylvaleric acid
- A-Oxo-b-methyl-N-valerate
- A-Oxo-b-methyl-N-valeric acid
- A-Oxo-b-methylvalerate
- A-Oxo-b-methylvaleric acid
- Alpha-Keto-beta-methyl-N-valerate
- Alpha-Keto-beta-methyl-N-valeric acid
- Alpha-Keto-beta-methyl-valerate
- Alpha-Keto-beta-methyl-valeric acid
- Alpha-Keto-beta-methylvalerate
- Alpha-Keto-beta-methylvaleric acid
- Alpha-Keto-methylvalerate
- Alpha-Keto-methylvaleric acid
- Alpha-Oxo-beta-methyl-N-valerate
- Alpha-Oxo-beta-methyl-N-valeric acid
- Alpha-Oxo-beta-methylvalerate
- Alpha-Oxo-beta-methylvaleric acid
- α-keto-Methylvalerate
- α-keto-Methylvaleric acid
- α-keto-β-Methyl-N-valerate
- α-keto-β-Methyl-N-valeric acid
- α-keto-β-Methyl-valerate
- α-keto-β-Methyl-valeric acid
- α-keto-β-Methylvalerate
- α-keto-β-Methylvaleric acid
- α-oxo-β-Methyl-N-valerate
- α-oxo-β-Methyl-N-valeric acid
- α-oxo-β-Methylvalerate
- α-oxo-β-Methylvaleric acid
|
|---|
|
Chemical Formula: |
C6H9O3 |
|---|
| Average Molecular Weight: |
129.1339 |
|---|
| Monoisotopic Molecular
Weight: |
129.055169154 |
|---|
| InChI Key: |
JVQYSWDUAOAHFM-BYPYZUCNSA-M |
|---|
| InChI: | InChI=1S/C6H10O3/c1-3-4(2)5(7)6(8)9/h4H,3H2,1-2H3,(H,8,9)/p-1/t4-/m0/s1 |
|---|
| CAS
number: |
1460-34-0 |
|---|
| IUPAC Name: | (3S)-3-methyl-2-oxopentanoate |
|---|
|
Traditional IUPAC Name: |
(S)-3-methyl-2-oxopentanoate |
|---|
| SMILES: | CC[C@H](C)C(=O)C([O-])=O |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as methyl-branched fatty acids. These are fatty acids with an acyl chain that has a methyl branch. Usually, they are saturated and contain only one or more methyl group. However, branches other than methyl may be present. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Fatty Acyls |
|---|
| Sub Class | Fatty acids and conjugates |
|---|
|
Direct Parent |
Methyl-branched fatty acids |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Methyl-branched fatty acid
- Keto acid
- Alpha-keto acid
- Ketone
- Carboxylic acid salt
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Organic anion
- Aliphatic acyclic compound
|
|---|
| Molecular Framework |
Aliphatic acyclic compounds |
|---|
| External Descriptors |
|
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -1 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
- Valine, leucine and isoleucine biosynthesis pae00290
- Valine, leucine and isoleucine degradation pae00280
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- van der Werf, M. J., Overkamp, K. M., Muilwijk, B., Coulier, L., Hankemeier, T. (2007). "Microbial metabolomics: toward a platform with full metabolome coverage." Anal Biochem 370:17-25. Pubmed: 17765195
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|