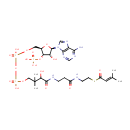|
Record Information |
|---|
| Version |
1.0 |
|---|
| Update Date |
1/22/2018 12:54:54 PM |
|---|
|
Metabolite ID | PAMDB000401 |
|---|
|
Identification |
|---|
| Name: |
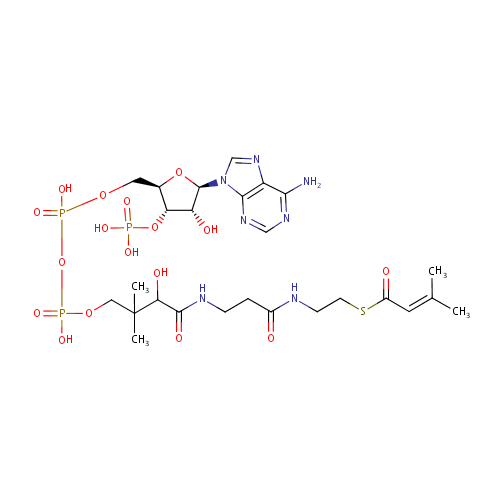
3-Methylcrotonyl-CoA |
|---|
| Description: | 3-Methylcrotonyl-CoA is an essential metabolite for leucine metabolism. |
|---|
|
Structure |
|
|---|
| Synonyms: | - β-methylcrotonoyl-CoA
- 3-Methylbut-2-enoyl-CoA
- 3-Methylbut-2-enoyl-Coenzyme A
- 3-Methylcrotonoyl-CoA
- 3-Methylcrotonoyl-Coenzyme A
- 3-Methylcrotonyl-CoA
- 3-Methylcrotonyl-Coenzyme A
- b-Methylcrotonoyl-CoA
- b-Methylcrotonoyl-coenzyme A
- b-Methylcrotonyl-CoA
- B-Methylcrotonyl-CoA (HMDB01493
- B-Methylcrotonyl-Coenzyme A
- B-Methylcrotonyl-Coenzyme A (HMDB01493
- Beta-Methylcrotonoyl-CoA
- Beta-Methylcrotonoyl-Coenzyme A
- Beta-Methylcrotonyl-CoA
- Beta-Methylcrotonyl-CoA (HMDB01493
- Beta-Methylcrotonyl-Coenzyme A
- Beta-Methylcrotonyl-Coenzyme A (HMDB01493
- Dimethylacryloyl-CoA
- Dimethylacryloyl-Coenzyme A
- β-Methylcrotonoyl-CoA
- β-Methylcrotonoyl-coenzyme A
- β-Methylcrotonyl-CoA
- β-Methylcrotonyl-CoA (HMDB01493
- β-Methylcrotonyl-coenzyme A
- β-Methylcrotonyl-coenzyme A (HMDB01493
|
|---|
|
Chemical Formula: |
C26H42N7O17P3S |
|---|
| Average Molecular Weight: |
849.635 |
|---|
| Monoisotopic Molecular
Weight: |
849.157073179 |
|---|
| InChI Key: |
BXIPALATIYNHJN-TVCSPYKZSA-N |
|---|
| InChI: | InChI=1S/C26H42N7O17P3S/c1-14(2)9-17(35)54-8-7-28-16(34)5-6-29-24(38)21(37)26(3,4)11-47-53(44,45)50-52(42,43)46-10-15-20(49-51(39,40)41)19(36)25(48-15)33-13-32-18-22(27)30-12-31-23(18)33/h9,12-13,15,19-21,25,36-37H,5-8,10-11H2,1-4H3,(H,28,34)(H,29,38)(H,42,43)(H,44,45)(H2,27,30,31)(H2,39,40,41)/t15-,19-,20-,21?,25-/m1/s1 |
|---|
| CAS
number: |
793193-48-3 |
|---|
| IUPAC Name: | {[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-4-hydroxy-2-({[hydroxy({[hydroxy(3-hydroxy-2,2-dimethyl-3-{[2-({2-[(3-methylbut-2-enoyl)sulfanyl]ethyl}carbamoyl)ethyl]carbamoyl}propoxy)phosphoryl]oxy})phosphoryl]oxy}methyl)oxolan-3-yl]oxy}phosphonic acid |
|---|
|
Traditional IUPAC Name: |
[(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-4-hydroxy-2-[({hydroxy[hydroxy(3-hydroxy-2,2-dimethyl-3-{[2-({2-[(3-methylbut-2-enoyl)sulfanyl]ethyl}carbamoyl)ethyl]carbamoyl}propoxy)phosphoryl]oxyphosphoryl}oxy)methyl]oxolan-3-yl]oxyphosphonic acid |
|---|
| SMILES: | CC(C)=CC(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C1N=CN=C2N |
|---|
|
Chemical Taxonomy |
|---|
|
Taxonomy Description | This compound belongs to the class of organic compounds known as acyl coas. These are organic compounds containing a coenzyme A substructure linked to an acyl chain. |
|---|
|
Kingdom |
Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
|
Class |
Fatty Acyls |
|---|
| Sub Class | Fatty acyl thioesters |
|---|
|
Direct Parent |
Acyl CoAs |
|---|
| Alternative Parents |
|
|---|
| Substituents |
- Coenzyme a or derivatives
- Purine ribonucleoside diphosphate
- Purine ribonucleoside 3',5'-bisphosphate
- N-glycosyl compound
- Glycosyl compound
- Beta amino acid or derivatives
- Organic pyrophosphate
- Monosaccharide phosphate
- 6-aminopurine
- Purine
- Imidazopyrimidine
- Monoalkyl phosphate
- Aminopyrimidine
- Imidolactam
- Alkyl phosphate
- Pyrimidine
- Primary aromatic amine
- Phosphoric acid ester
- Organic phosphoric acid derivative
- Organic phosphate
- N-substituted imidazole
- N-acyl-amine
- Monosaccharide
- Fatty amide
- Heteroaromatic compound
- Oxolane
- Imidazole
- Azole
- Thiocarboxylic acid ester
- Secondary carboxylic acid amide
- Secondary alcohol
- Carboxamide group
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Sulfenyl compound
- Thioether
- Thiocarboxylic acid or derivatives
- Carboxylic acid derivative
- Carboxylic acid amide
- Hydrocarbon derivative
- Primary amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Amine
- Alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework |
Aromatic heteropolycyclic compounds |
|---|
| External Descriptors |
Not Available |
|---|
|
Physical Properties |
|---|
| State: |
Solid |
|---|
| Charge: | -4 |
|---|
|
Melting point: |
Not Available |
|---|
| Experimental Properties: |
|
|---|
| Predicted Properties |
|
|---|
|
Biological Properties |
|---|
| Cellular Locations: |
Cytoplasm |
|---|
| Reactions: | |
|---|
|
Pathways: |
- Geraniol degradation pae00281
- Valine, leucine and isoleucine degradation pae00280
|
|---|
|
Spectra |
|---|
| Spectra: |
|
|---|
|
References |
|---|
| References: |
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- van der Werf, M. J., Overkamp, K. M., Muilwijk, B., Coulier, L., Hankemeier, T. (2007). "Microbial metabolomics: toward a platform with full metabolome coverage." Anal Biochem 370:17-25. Pubmed: 17765195
- Winder, C. L., Dunn, W. B., Schuler, S., Broadhurst, D., Jarvis, R., Stephens, G. M., Goodacre, R. (2008). "Global metabolic profiling of Escherichia coli cultures: an evaluation of methods for quenching and extraction of intracellular metabolites." Anal Chem 80:2939-2948. Pubmed: 18331064
- Wysocki SJ, Hahnel R: 3-Methylcrotonylglycine excretion in 3-hydroxy-3-methylglutaric aciduria. Clin Chim Acta. 1978 May 16;86(1):101-8. Pubmed: 657530
|
|---|
| Synthesis Reference: |
Not Available |
|---|
| Material Safety Data Sheet (MSDS) |
Not Available |
|---|
|
Links |
|---|
| External Links: |
|
|---|