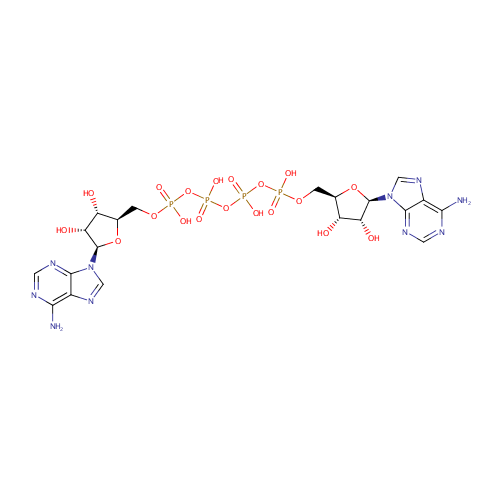
| InChI: | InChI=1S/C20H28N10O19P4/c21-15-9-17(25-3-23-15)29(5-27-9)19-13(33)11(31)7(45-19)1-43-50(35,36)47-52(39,40)49-53(41,42)48-51(37,38)44-2-8-12(32)14(34)20(46-8)30-6-28-10-16(22)24-4-26-18(10)30/h3-8,11-14,19-20,31-34H,1-2H2,(H,35,36)(H,37,38)(H,39,40)(H,41,42)(H2,21,23,25)(H2,22,24,26)/t7-,8-,11-,12-,13-,14-,19-,20-/m1/s1 |
|---|
| References: |
- Baker MD, Holloway DE, Swaminathan GJ, Acharya KR: Crystal structures of eosinophil-derived neurotoxin (EDN) in complex with the inhibitors 5'-ATP, Ap3A, Ap4A, and Ap5A. Biochemistry. 2006 Jan 17;45(2):416-26. Pubmed: 16401072
- Chan PJ, Su BC, Tredway DR: Diadenosine tetraphosphate (Ap4A) and triphosphate (Ap3A) signaling of human sperm motility. Arch Androl. 1991 Sep-Oct;27(2):103-8. Pubmed: 1953194
- de Korte D, Gouwerok CW, Fijnheer R, Pietersz RN, Roos D: Depletion of dense granule nucleotides during storage of human platelets. Thromb Haemost. 1990 Apr 12;63(2):275-8. Pubmed: 2141956
- Gualix J, Pintor J, Miras-Portugal MT: Characterization of nucleotide transport into rat brain synaptic vesicles. J Neurochem. 1999 Sep;73(3):1098-104. Pubmed: 10461900
- Hollah P, Hausberg M, Kosch M, Barenbrock M, Letzel M, Schlatter E, Rahn KH: A novel assay for determination of diadenosine polyphosphates in human platelets: studies in normotensive subjects and in patients with essential hypertension. J Hypertens. 2001 Feb;19(2):237-45. Pubmed: 11212966
- Hourani SM, Bailey SJ, Johnson CR, Tennant JP: Effects of adenosine 5'-triphosphate, uridine 5'-triphosphate, adenosine 5'-tetraphosphate and diadenosine polyphosphates in guinea-pig taenia caeci and rat colon muscularis mucosae. Naunyn Schmiedebergs Arch Pharmacol. 1998 Oct;358(4):464-73. Pubmed: 9826069
- Hoyle CH: Pharmacological activity of adenine dinucleotides in the periphery: possible receptor classes and transmitter function. Gen Pharmacol. 1990;21(6):827-31. Pubmed: 2279683
- Jankowski J, Jankowski V, Laufer U, van der Giet M, Henning L, Tepel M, Zidek W, Schluter H: Identification and quantification of diadenosine polyphosphate concentrations in human plasma. Arterioscler Thromb Vasc Biol. 2003 Jul 1;23(7):1231-8. Epub 2003 May 8. Pubmed: 12738682
- Jankowski J, Schluter H, Tepel M, Spieker C, Zidek W: Effect of diadenosine polyphosphates on Ca2+ ATPase activity. J Mol Med. 1997 Sep;75(9):674-7. Pubmed: 9351706
- Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., Tanabe, M. (2012). "KEGG for integration and interpretation of large-scale molecular data sets." Nucleic Acids Res 40:D109-D114. Pubmed: 22080510
- Keseler, I. M., Collado-Vides, J., Santos-Zavaleta, A., Peralta-Gil, M., Gama-Castro, S., Muniz-Rascado, L., Bonavides-Martinez, C., Paley, S., Krummenacker, M., Altman, T., Kaipa, P., Spaulding, A., Pacheco, J., Latendresse, M., Fulcher, C., Sarker, M., Shearer, A. G., Mackie, A., Paulsen, I., Gunsalus, R. P., Karp, P. D. (2011). "EcoCyc: a comprehensive database of Escherichia coli biology." Nucleic Acids Res 39:D583-D590. Pubmed: 21097882
- Kisselev LL, Justesen J, Wolfson AD, Frolova LY: Diadenosine oligophosphates (Ap(n)A), a novel class of signalling molecules? FEBS Lett. 1998 May 8;427(2):157-63. Pubmed: 9607303
- Pintor J, Carracedo G, Alonso MC, Bautista A, Peral A: Presence of diadenosine polyphosphates in human tears. Pflugers Arch. 2002 Jan;443(3):432-6. Epub 2001 Aug 23. Pubmed: 11810214
- Pintor J, King BF, Miras-Portugal MT, Burnstock G: Selectivity and activity of adenine dinucleotides at recombinant P2X2 and P2Y1 purinoceptors. Br J Pharmacol. 1996 Nov;119(5):1006-12. Pubmed: 8922753
- Pintor J, Puche JA, Gualix J, Hoyle CH, Miras-Portugal MT: Diadenosine polyphosphates evoke Ca2+ transients in guinea-pig brain via receptors distinct from those for ATP. J Physiol. 1997 Oct 15;504 ( Pt 2):327-35. Pubmed: 9365907
- Pirrone AM, Gambino R, Oddo F, Faraci MT, Luparello G, Giudice G: Sea urchin embryos do not synthesize diadenosinetetraphosphate. Exp Cell Res. 1979 Sep;122(2):419-22. Pubmed: 510412
- Stachon A, Stegemann H, Hohage H, Rahn KH, Schlatter E: Effects of diadenosine polyphosphates on the intracellular Ca2+ concentration in endothelial cells. Cell Physiol Biochem. 1998;8(4):175-84. Pubmed: 9694344
- Stiepanow-Trzeciak A, Jankowski M, Angielski S, Szczepanska-Konkel M: P1,P4-diadenosine tetraphosphate (Ap4A) inhibits proximal tubular reabsorption of sodium in rats. Nephron Physiol. 2007;106(1):p13-8. Epub 2007 Apr 2. Pubmed: 17406124
- Turpaev K, Hartmann R, Kisselev L, Justesen J: Ap3A and Ap4A are primers for oligoadenylate synthesis catalyzed by interferon-inducible 2-5A synthetase. FEBS Lett. 1997 May 19;408(2):177-81. Pubmed: 9187362
- van der Giet M, Jankowski J, Schluter H, Zidek W, Tepel M: Mediation of the vasoactive properties of diadenosine tetraphosphate via various purinoceptors. J Hypertens. 1998 Dec;16(12 Pt 2):1939-43. Pubmed: 9886880
- van der Giet M, Khattab M, Borgel J, Schluter H, Zidek W: Differential effects of diadenosine phosphates on purinoceptors in the rat isolated perfused kidney. Br J Pharmacol. 1997 Apr;120(8):1453-60. Pubmed: 9113365
- Wildman SS, Brown SG, King BF, Burnstock G: Selectivity of diadenosine polyphosphates for rat P2X receptor subunits. Eur J Pharmacol. 1999 Feb 12;367(1):119-23. Pubmed: 10082274
- Winder, C. L., Dunn, W. B., Schuler, S., Broadhurst, D., Jarvis, R., Stephens, G. M., Goodacre, R. (2008). "Global metabolic profiling of Escherichia coli cultures: an evaluation of methods for quenching and extraction of intracellular metabolites." Anal Chem 80:2939-2948. Pubmed: 18331064
|
|---|