4-Phosphopantothenoylcysteine (PAMDB000259)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 1/22/2018 12:54:54 PM | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolite ID | PAMDB000259 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
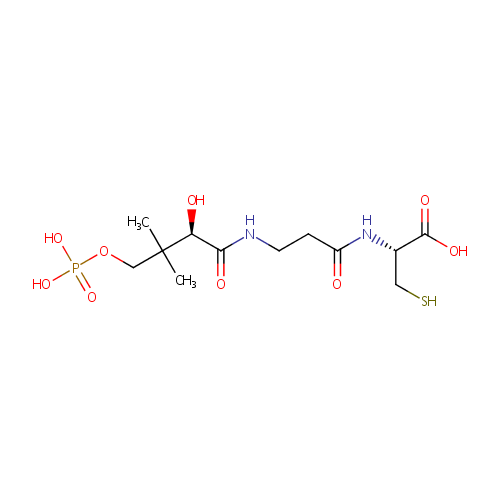
| Name: | 4-Phosphopantothenoylcysteine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description: | 4-Phosphopantothenoylcysteine (PPC) is an intermediate in the biosynthetic machinery (pathway) that converts pantothenate (vitamin B5) into coenzyme A (CoA). The enzyme Phosphopantothenoylcysteine decarboxylase catalyzes the decarboxylation of PPC to 4'-phosphopantetheine. Coenzyme A is the principal acyl carrier and is required for many synthetic and degradative reactions in intermediary metabolism, and is an essential cofactor in all living systems. (PMID: 15450493, 16371361, 14501115) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula: | C12H23N2O9PS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Weight: | 402.358 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Molecular Weight: | 402.086187546 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key: | XQYALQVLCNHCFT-CBAPKCEASA-N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI: | InChI=1S/C12H23N2O9PS/c1-12(2,6-23-24(20,21)22)9(16)10(17)13-4-3-8(15)14-7(5-25)11(18)19/h7,9,16,25H,3-6H2,1-2H3,(H,13,17)(H,14,15)(H,18,19)(H2,20,21,22)/t7-,9-/m0/s1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number: | 7196-09-0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name: | (2R)-2-{3-[(2R)-2-hydroxy-3-methyl-3-[(phosphonooxy)methyl]butanamido]propanamido}-3-sulfanylpropanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional IUPAC Name: | (2R)-2-{3-[(2R)-2-hydroxy-3-methyl-3-[(phosphonooxy)methyl]butanamido]propanamido}-3-sulfanylpropanoic acid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES: | CC(C)(COP(O)(O)=O)[C@@H](O)C(=O)NCCC(=O)N[C@@H](CS)C(O)=O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taxonomy Description | This compound belongs to the class of organic compounds known as hybrid peptides. These are compounds containing at least two different types of amino acids (alpha, beta, gamma, delta) linked to each other through a peptide bond. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Organic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Carboxylic acids and derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Amino acids, peptides, and analogues | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Hybrid peptides | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Aliphatic acyclic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State: | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charge: | -3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations: | Cytoplasm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reactions: | D-4'-Phosphopantothenate + Cytidine triphosphate + L-Cysteine > 4-Phosphopantothenoylcysteine + Cytidine monophosphate + Hydrogen ion + Pyrophosphate 4-Phosphopantothenoylcysteine + Hydrogen ion <> Carbon dioxide + Pantetheine 4'-phosphate + pantotheine 4'-phosphate 4-Phosphopantothenoylcysteine <> Pantetheine 4'-phosphate + Carbon dioxide Adenosine triphosphate + D-4'-Phosphopantothenate + L-Cysteine <> Adenosine monophosphate + Pyrophosphate + 4-Phosphopantothenoylcysteine Cytidine triphosphate + D-4'-Phosphopantothenate + L-Cysteine <> Cytidine monophosphate + Pyrophosphate + 4-Phosphopantothenoylcysteine Adenosine triphosphate + D-Pantothenoyl-L-cysteine <> ADP + 4-Phosphopantothenoylcysteine Hydrogen ion + 4-Phosphopantothenoylcysteine > Pantetheine 4'-phosphate + Carbon dioxide 4-Phosphopantothenoylcysteine > pantotheine 4'-phosphate + Carbon dioxide Adenosine triphosphate + D-Pantothenoyl-L-cysteine > Adenosine diphosphate + 4-Phosphopantothenoylcysteine + ADP Cytidine triphosphate + D-4'-Phosphopantothenate + L-Cysteine + D-4'-Phosphopantothenate > Cytidine monophosphate + Pyrophosphate + 4-Phosphopantothenoylcysteine + Cytidine monophosphate 4-Phosphopantothenoylcysteine > Pantetheine 4'-phosphate + Carbon dioxide + pantotheine 4'-phosphate D-4'-Phosphopantothenate + Cytidine triphosphate + L-Cysteine + D-4'-Phosphopantothenate > Cytidine monophosphate + Pyrophosphate + Hydrogen ion + 4-Phosphopantothenoylcysteine + Cytidine monophosphate 4-Phosphopantothenoylcysteine + Hydrogen ion > Carbon dioxide + 4'-phosphopantetheine + 4'-phosphopantetheine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference: | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet (MSDS) | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links: |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||