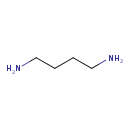
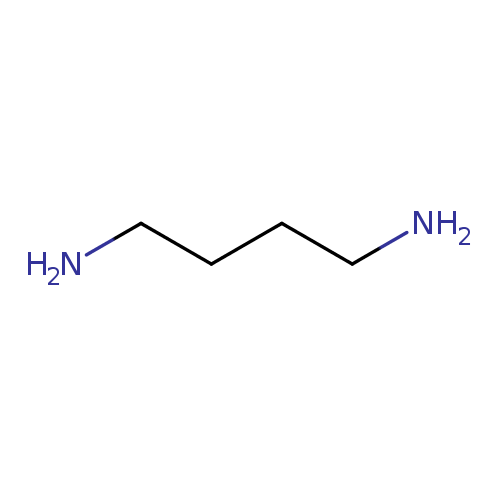
| References: |
- Gimelli G, Giglio S, Zuffardi O, Alhonen L, Suppola S, Cusano R, Lo Nigro C, Gatti R, Ravazzolo R, Seri M: Gene dosage of the spermidine/spermine N(1)-acetyltransferase ( SSAT) gene with putrescine accumulation in a patient with a Xp21.1p22.12 duplication and keratosis follicularis spinulosa decalvans (KFSD). Hum Genet. 2002 Sep;111(3):235-41. Epub 2002 Aug 1. [12215835 ]
- Janne J, Alhonen L, Pietila M, Keinanen TA: Genetic approaches to the cellular functions of polyamines in mammals. Eur J Biochem. 2004 Mar;271(5):877-94. [15009201 ]
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. [19212411 ]
- Venza M, Visalli M, Cicciu D, Teti D: Determination of polyamines in human saliva by high-performance liquid chromatography with fluorescence detection. J Chromatogr B Biomed Sci Appl. 2001 Jun 5;757(1):111-7. [11419735 ]
- El Baze P, Milano G, Verrando P, Renee N, Ortonne JP: Polyamine levels in normal human skin. A comparative study of pure epidermis, pure dermis, and suction blister fluid. Arch Dermatol Res. 1983;275(4):218-21. [6625645 ]
- Reeben M, Arbatova J, Palgi J, Miettinen R, Halmekyto M, Alhonen L, Janne J, Riekkinen P Sr, Saarma M: Induced expression of neurotrophins in transgenic mice overexpressing ornithine decarboxylase and overproducing putrescine. J Neurosci Res. 1996 Sep 1;45(5):542-8. [8875319 ]
- Takagi K, Tatsumi Y, Kitaichi K, Iwase M, Shibata E, Nakao M, Matsumoto T, Takagi K, Hasegawa T: A sensitive colorimetric assay for polyamines in erythrocytes using oat seedling polyamine oxidase. Clin Chim Acta. 2004 Feb;340(1-2):219-27. [14734216 ]
- Harik SI, Sutton CH: Putrescine as a biochemical marker of malignant brain tumors. Cancer Res. 1979 Dec;39(12):5010-5. [227593 ]
- Halmekyto M, Alhonen L, Alakuijala L, Janne J: Transgenic mice over-producing putrescine in their tissues do not convert the diamine into higher polyamines. Biochem J. 1993 Apr 15;291 ( Pt 2):505-8. [8484731 ]
- Goldman SS, Volkow ND, Brodie J, Flamm ES: Putrescine metabolism in human brain tumors. J Neurooncol. 1986;4(1):23-9. [3746382 ]
- Yamazaki H, Tsukahara T, Uki J, Matsuzaki S: Elevated levels of free putrescine and N1-acetylspermidine in cyst fluids of malignant brain tumours. J Neurol Neurosurg Psychiatry. 1986 Feb;49(2):209-10. [3950641 ]
- Thiele I, Swainston N, Fleming RM, Hoppe A, Sahoo S, Aurich MK, Haraldsdottir H, Mo ML, Rolfsson O, Stobbe MD, Thorleifsson SG, Agren R, Bolling C, Bordel S, Chavali AK, Dobson P, Dunn WB, Endler L, Hala D, Hucka M, Hull D, Jameson D, Jamshidi N, Jonsson JJ, Juty N, Keating S, Nookaew I, Le Novere N, Malys N, Mazein A, Papin JA, Price ND, Selkov E Sr, Sigurdsson MI, Simeonidis E, Sonnenschein N, Smallbone K, Sorokin A, van Beek JH, Weichart D, Goryanin I, Nielsen J, Westerhoff HV, Kell DB, Mendes P, Palsson BO: A community-driven global reconstruction of human metabolism. Nat Biotechnol. 2013 Mar 3. doi: 10.1038/nbt.2488. [23455439 ]
- Janne J, Alhonen L, Keinanen TA, Pietila M, Uimari A, Pirinen E, Hyvonen MT, Jarvinen A: Animal disease models generated by genetic engineering of polyamine metabolism. J Cell Mol Med. 2005 Oct-Dec;9(4):865-82. [16364196 ]
|
|---|